To investigate the effects of injectable hormonal contraceptives on tear secretion and tear stability of females within child bearing age in Nigeria.
MethodsThe experimental group consisted of 32 healthy females (mean age was 34.72±5.44) on injectable hormonal contraceptives; and the control group comprised 32 females (mean age was 34.66±5.24) who were not on hormonal contraceptives. The tear stability and tear secretion were measured using the non-invasive tear break up time (NITBUT) technique and Schirmer's strips, respectively. All the females were at follicular phase of menstrual cycle. The plasma levels of progesterone and estradiol of all subjects were determined.
ResultsThere were no remarkable effects of injectable hormonal contraceptives on tear secretion (P=0.929) and tear stability (P=0.814). There were weak correlations between the plasma levels of progesterone and tear secretion (r=−0.232, P>0.05), as well as with tear stability (r=−0.322, P>0.05). Also, there were weak positive correlation between plasma levels of estradiol and tear secretion (r=0.304, P>0.05), as well as with tear stability (r=0.262, P>0.05). There were no significant differences in tear stability between the experimental and control groups (P>0.05).
ConclusionsInjectable hormonal contraceptives had no significant effects on tear secretion and tear stability of healthy women of childbearing age. Further studies may be required to determine the effects of hormonal contraceptives on tear volume and stability of women with dry eyes.
El estudio tuvo como objetivo la investigación de los efectos de los anticonceptivos hormonales inyectables sobre la secreción y la estabilidad de la lágrima en mujeres en edad fértil en Nigeria.
MétodosEl grupo experimental estaba compuesto por 32 mujeres sanas (edad media de 34,72 ± 5,44 años) sometidas a terapia anticonceptiva hormonal inyectable; y el grupo de control incluía a 32 mujeres (edad media de 34,66 ± 5,24 años) no sometidas a anticoncepción hormonal. La estabilidad y la secreción de la lágrima se midieron mediante la técnica NITBUT (tiempo de ruptura de la capa lipídica de la lágrima) y las tiras de Schirmer, respectivamente. Todas las mujeres se hallaban en la fase folicular del ciclo menstrual. Se determinaron los niveles de progesterona y de estradiol de todas las pacientes.
ResultadosNo se hallaron efectos destacables de los anticonceptivos hormonales inyectables sobre la secreción (P=0,929) y la estabilidad de la lágrima (P=0,814). Se produjeron correlaciones leves entre los niveles de plasma de la progesterona y la secreción de la lágrima (r=-0,232, P>0,05), y la estabilidad de la lágrima (r=-0,322, P>0,05). También se halló una débil correlación entre los niveles de plasma del estradiol y las secreción de la lágrima (r=0,304, P>0,05), y la estabilidad de la lágrima (r=0,262, P>0,05). No se produjeron diferencias significativas en cuanto a estabilidad de la lágrima entre los grupos experimental y de control (P > 0,05).
ConclusionesLos anticonceptivos hormonales inyectables no tienen efectos significativos sobre la secreción y la estabilidad de la lágrima en las mujeres sanas en edad fértil. Pueden precisarse estudios adicionales para determinar los efectos de los anticonceptivos hormonales sobre el volumen y la estabilidad de la lágrima en mujeres con ojo seco.
The endocrine system exerts significant influences on the physiology and pathophysiology of the lacrimal gland. Androgens, estrogens and progestin have been identified in the tear film and their levels in the tears appeared to correlate with those of the serum.1 Receptors for androgens, estrogens, progesterone and prolactin have been found in several ocular tissues of rats, rabbits and humans. These hormones regulate the immune system, secretary functions of lacrimal and the meibomian glands.2,3 Thus the eye is a target organ for sex hormones. Rocha et al.4 reported that androgen, estrogen and progesterone receptors mRNAs were present in the epithelial cells of the lacrimal gland, meibomian gland, lid, palpebral and bulbar conjunctivae, cornea, uveal body, lens, and retina of humans. These observations demonstrate that sex steroid receptors mRNAs exist in a variety of ocular tissues. It was suggested that these receptors in the eye might be target sites for androgens, estrogen and progestin; and that these sites might also be susceptible to administered topical and systemic hormonal contraceptives. Several authors reported that these sex steroids (i.e. androgens, estrogens and progestin) modulate the structural characteristics, functional attributes and pathological features of ocular tissues. These observations had accounted for the gender-related differences in dry eyes.5–7
Injectable contraceptives are hormone shots administered to some women in order to prevent pregnancy. An estimated 16 million women throughout the world use hormonal contraceptives.8 There are controversies on the effects of the injectable hormonal contraceptives on tear physiology and tear production of women. Sullivan et al.3 suggested that hormonal contraceptives improved the quality and the production of tear film, but other authors9 reported that the hormone increases the risk of dry eyes in women. Some researchers10–12 proposed also that either the topical or systemic application of estrogens might ameliorate the symptoms of dry eyes. There were other suggestions that oral contraceptives containing estrogen might decrease tear volume and reduce tear break-up time. It was also suspected that estrogen oral contraceptives might attenuate mucous production, increase foreign body sensation, reduce contact lens intolerance, decrease visual acuity and increase the risk of dry eyes in women using the drugs.3,13,14 It has been recorded that post-menopausal women under hormone replacement therapy (HRT), particularly estrogen alone, were at risk of dry eyes syndrome.9 These controversies prompted this study, aimed at investigating the effects of injectable contraceptives on tear secretion and stability of women within child bearing ages.
MethodsThe study was a prospective study carried out in the Department of Optometry, University of Benin, Benin City, Nigeria. The participants comprised sixty four (64) healthy women of child bearing ages, within the age bracket of 26–45 years. They were healthy female volunteers from the family planning clinic, Department of Obstetrics and Gynaecology, University of Benin teaching hospital, Benin City, Nigeria. The subjects consisted of 32 females, who were current users of injectable contraceptives. The control group consisted of 32 females who had not used any form of hormonal contraceptives. The control and the experimental groups were properly matched for age (within 3 months to one year differences in age) and the phase of menstrual cycle. All the subjects were Nigerians living in Edo and Delta states of Nigeria. Informed and verbal consents were obtained from each of the participants and the study was conducted in accordance with the tenants of the Declaration of Helsinki.
Questionnaires were administered to the subjects in order to obtain information about their personal data, the use of injectable contraceptives and other forms of contraceptives, the type of contraceptive used, duration of use, oculo-visual history, and general health history. Oral interviews were carried out for subjects who were not able to fill out the questionnaires, which were filled for them. A brief case history was obtained from each subject. The subjects’ ocular health and general health history were obtained. Visual acuity test was carried out using the standard Snellen's chart. External eye examination was carried out, using the slit lamp biomicroscope to rule out ocular surface and anterior segment abnormalities. The internal structures of the eyes were properly examined with a direct ophthalmoscope to rule out diseases of the posterior segment. Blood pressure was measured with the use of a sphygmomanometer to rule out hypertensive patients from the study. Participants who met the inclusion criteria were either put in the experimental group, if they were users of injectable hormonal contraceptives or in the control groups, and if they were not on hormonal contraceptives. These subjects were identified with serial numbers.
Inclusion criteria for the experimental and the control groupsWomen of child bearing age, within 26–45 years of age, who were only on injectable birth control hormones (such as Depo-Provera: 150mg depot medroxyprogesterone acetate, Noristerat: 200mg noresthisterone enanthate ‘NET-EN’) for at least a period of six months and in the follicular phase of menstrual cycle were enrolled in the experimental group. The control group consisted of females between 26 and 45 years of age who were either not on contraceptives or were not on hormonal contraceptives. All the female participants were in the follicular phase of the menstrual cycle.
Exclusion criteriaWomen with lid-gland dysfunction (blepharitis) ocular surface abnormalities and any other obvious ocular pathology were excluded from the study. Women with history of any systemic disease, ocular surgery, laser therapy and on any medications were not included in the study. Contact lenses wearers, pregnant women, smokers, women under topical eye drops, menopausal and post-menopausal women were also excluded.
Measurement of tear secretionTotal tear secretion (without anesthetic) was measured with Schirmer's test strip on the subject's right eye, which was gently dried to remove any excess tears. The subject was comfortably seated and was instructed to look up, while the lower lid was gently pulled down. The bent hooked end of the Schirmer's strip was then placed at the junction of the middle and outer two third of the lower lid, with care not to touch the cornea or lashes. The subject was asked to continuously look up and blink normally or to close the eye if it was more comfortable, but not to squeeze the eye. The strip was removed 5min after insertion. The wet portion of the strip from the bent notch was measured in mm and recorded. Three readings were taken for the sake of reliability with 10min interval between readings. The average of these readings was recorded for each subject and any value less than 10mm of wetting after 5min was considered abnormal.
Measurement of tear stabilityThe NITBUT was employed in the assessment of tear film stability for both eyes. When the subject and the examiner were comfortably seated, the subject was asked to look through the eyepiece, while necessary adjustments on the keratometer were carried out until the mires were visible. The subject was instructed to blink and to keep the eye open. The time between the last blink and the doubling or distortion of the mires was recorded with a stopwatch. The mean value for three readings was recorded for the NITBUT and dry eye was suspected for any value lesser than 10s.
Blood sample collectionBlood samples were collected into labeled (with serial number for identification) specimen tubes (without anticoagulant) and they were left to stand at room temperature (37°C) for 1h. The blood was then centrifuged at full speed in a micro centrifuge. The supernatant was removed and placed in a new specimen tube. The serum was stored at −20°C (for long term storage) prior to the analysis.
Hormonal assayThe plasma levels of progesterone and estrogen (estradiol) were assessed using Microwell Progesterone EIA (REF-4210-96) and Microwell Estradiol EIA (REF-5026-70), respectively.
Statistical analysisThe data was analyzed with STATGRAPHICS plus 5.1, Student's unpaired t-test was used to determine the effect of injectable contraceptive on tear secretion in the control and experimental groups and tear stability in the control and experimental groups. Pearson's correlation analysis and regression was used to determine the correlations between plasma levels of estrogen and progesterone with the tear secretion and tear stability. The results were significant if the probability values revealed P<0.05.
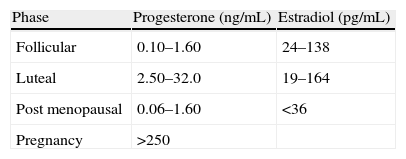
ResultsSerum levels of progesterone and estradiolThe expected normal values of progesterone and estradiol are shown in Table 1.
The mean serum levels of progesterone and estradiol obtained in this study were within expected values given in Table 1.
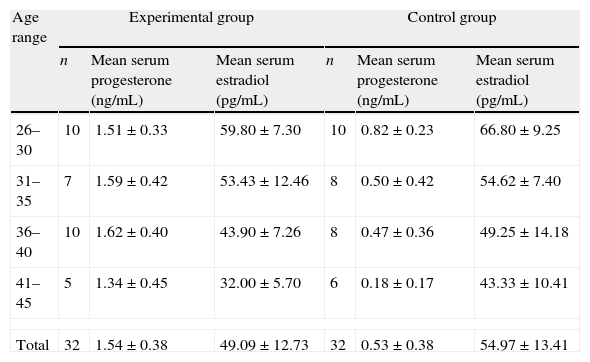
Table 2 shows that there were significantly higher serum progesterone levels in the experimental group than was found in the control group (P<0.05).
Mean levels of progesterone and estradiol.
| Age range | Experimental group | Control group | ||||
| n | Mean serum progesterone (ng/mL) | Mean serum estradiol (pg/mL) | n | Mean serum progesterone (ng/mL) | Mean serum estradiol (pg/mL) | |
| 26–30 | 10 | 1.51±0.33 | 59.80±7.30 | 10 | 0.82±0.23 | 66.80±9.25 |
| 31–35 | 7 | 1.59±0.42 | 53.43±12.46 | 8 | 0.50±0.42 | 54.62±7.40 |
| 36–40 | 10 | 1.62±0.40 | 43.90±7.26 | 8 | 0.47±0.36 | 49.25±14.18 |
| 41–45 | 5 | 1.34±0.45 | 32.00±5.70 | 6 | 0.18±0.17 | 43.33±10.41 |
| Total | 32 | 1.54±0.38 | 49.09±12.73 | 32 | 0.53±0.38 | 54.97±13.41 |
n: number.
The mean estradiol level in the control group was higher than that of the experimental group, but there were no statistically significant differences between these values (P>0.05).
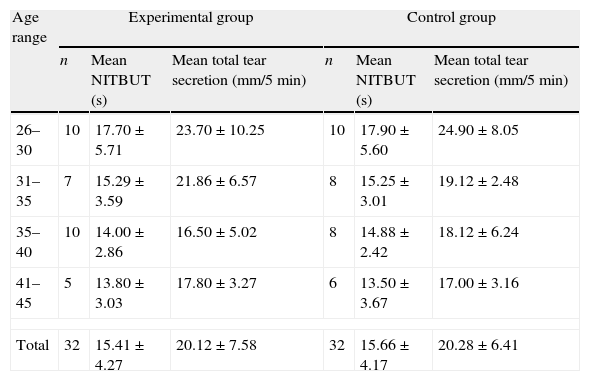
Tear secretionThe mean total tears secretion in mm per 5min in the experimental and control groups were 20.12±7.58mm/5min and 20.28±6.41mm/5min, respectively (Table 3). There were no significant differences in these values (P=0.929), using unpaired t-test.
Mean values of tear secretion and tear stability in different age groups.
| Age range | Experimental group | Control group | ||||
| n | Mean NITBUT (s) | Mean total tear secretion (mm/5min) | n | Mean NITBUT (s) | Mean total tear secretion (mm/5min) | |
| 26–30 | 10 | 17.70±5.71 | 23.70±10.25 | 10 | 17.90±5.60 | 24.90±8.05 |
| 31–35 | 7 | 15.29±3.59 | 21.86±6.57 | 8 | 15.25±3.01 | 19.12±2.48 |
| 35–40 | 10 | 14.00±2.86 | 16.50±5.02 | 8 | 14.88±2.42 | 18.12±6.24 |
| 41–45 | 5 | 13.80±3.03 | 17.80±3.27 | 6 | 13.50±3.67 | 17.00±3.16 |
| Total | 32 | 15.41±4.27 | 20.12±7.58 | 32 | 15.66±4.17 | 20.28±6.41 |
n: number.
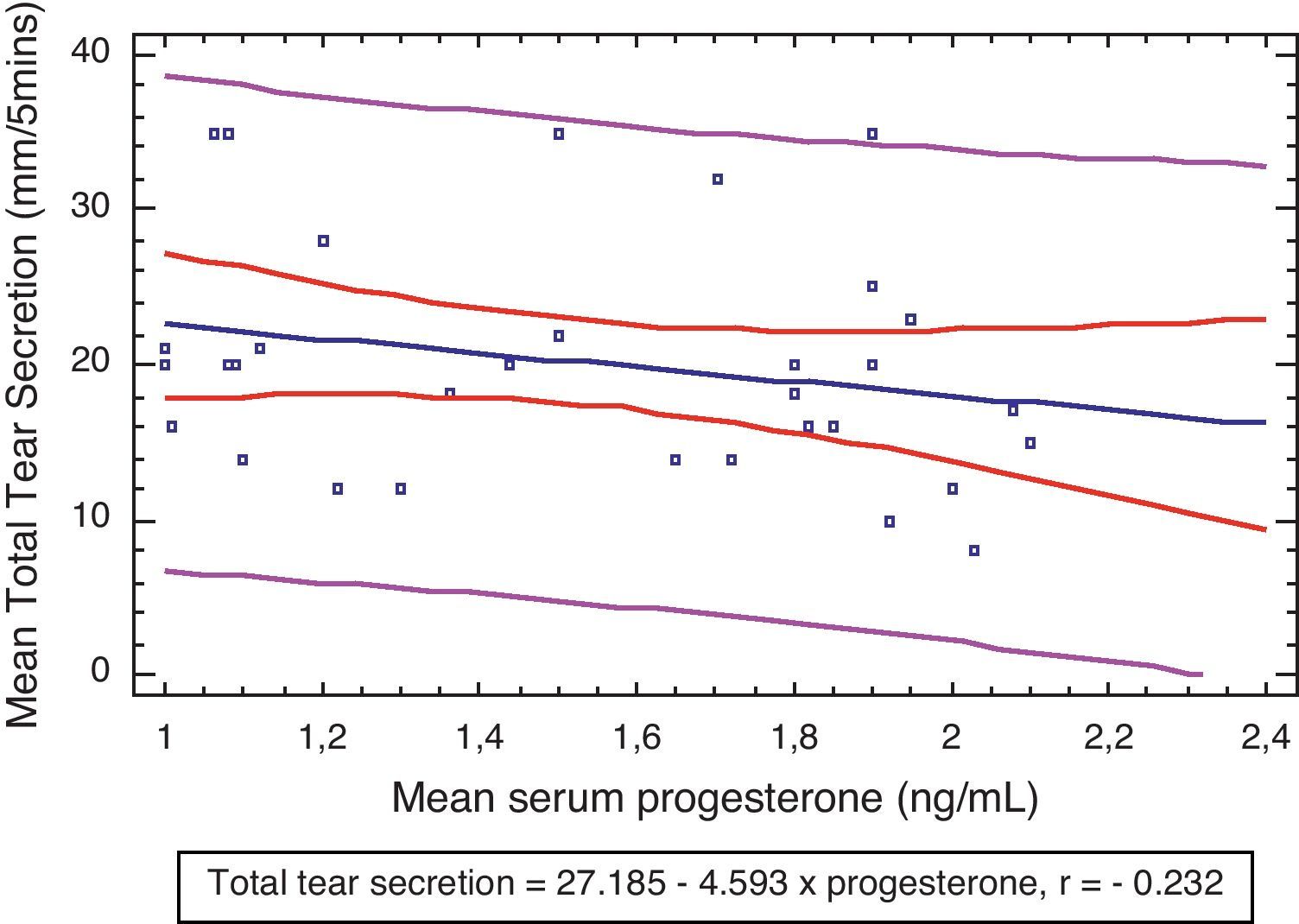
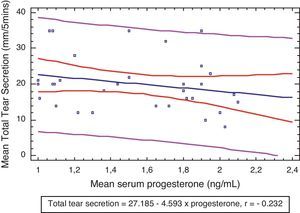
Fig. 1 shows that there were weak correlations between the plasma levels of progesterone and tear secretion (r=−0.232), although this was not statistically significant (P>0.05).
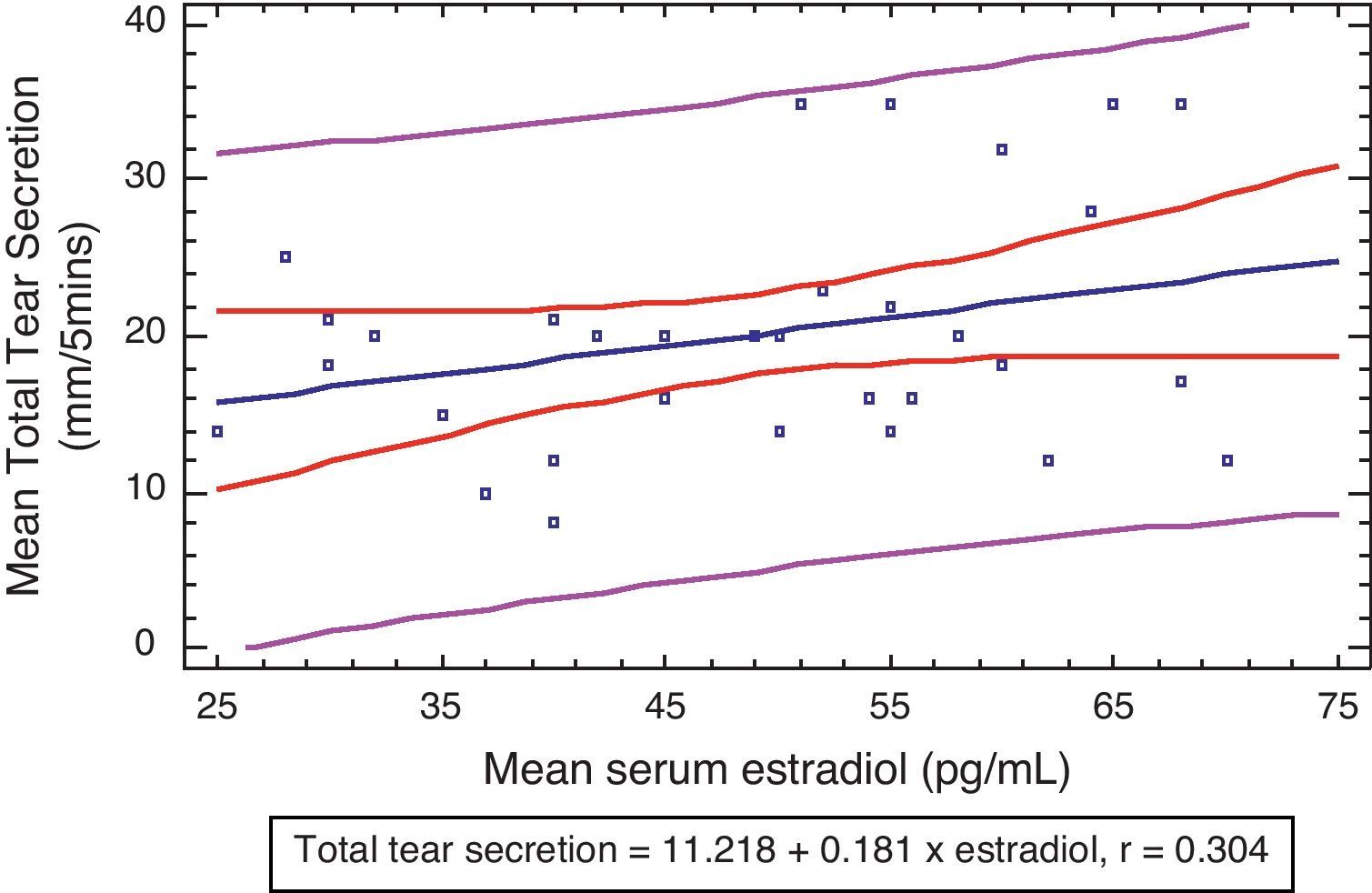
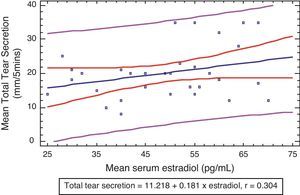
There were weak positive correlations between the plasma levels of estradiol and tear secretion (r=0.304) although this was also not statistically significant (P>0.05) (Fig. 2).
Tear stabilityThe mean NITBUT for experimental and control groups were 15.41±4.27s and 15.66±4.17s, respectively.
The unpaired t-test revealed also that there were no significant differences between the NITBUT for experimental and control groups (P=0.814) (Table 3).
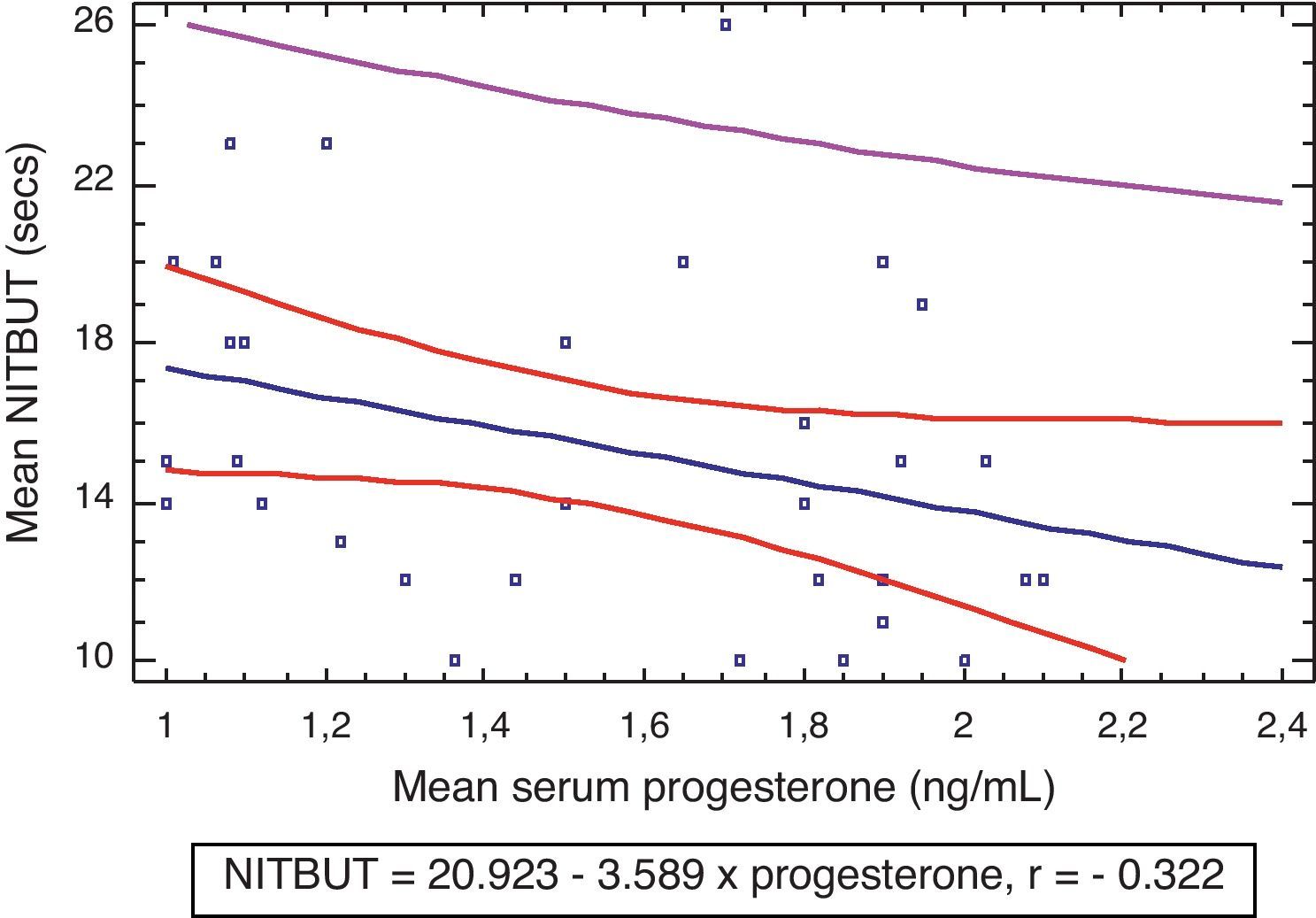
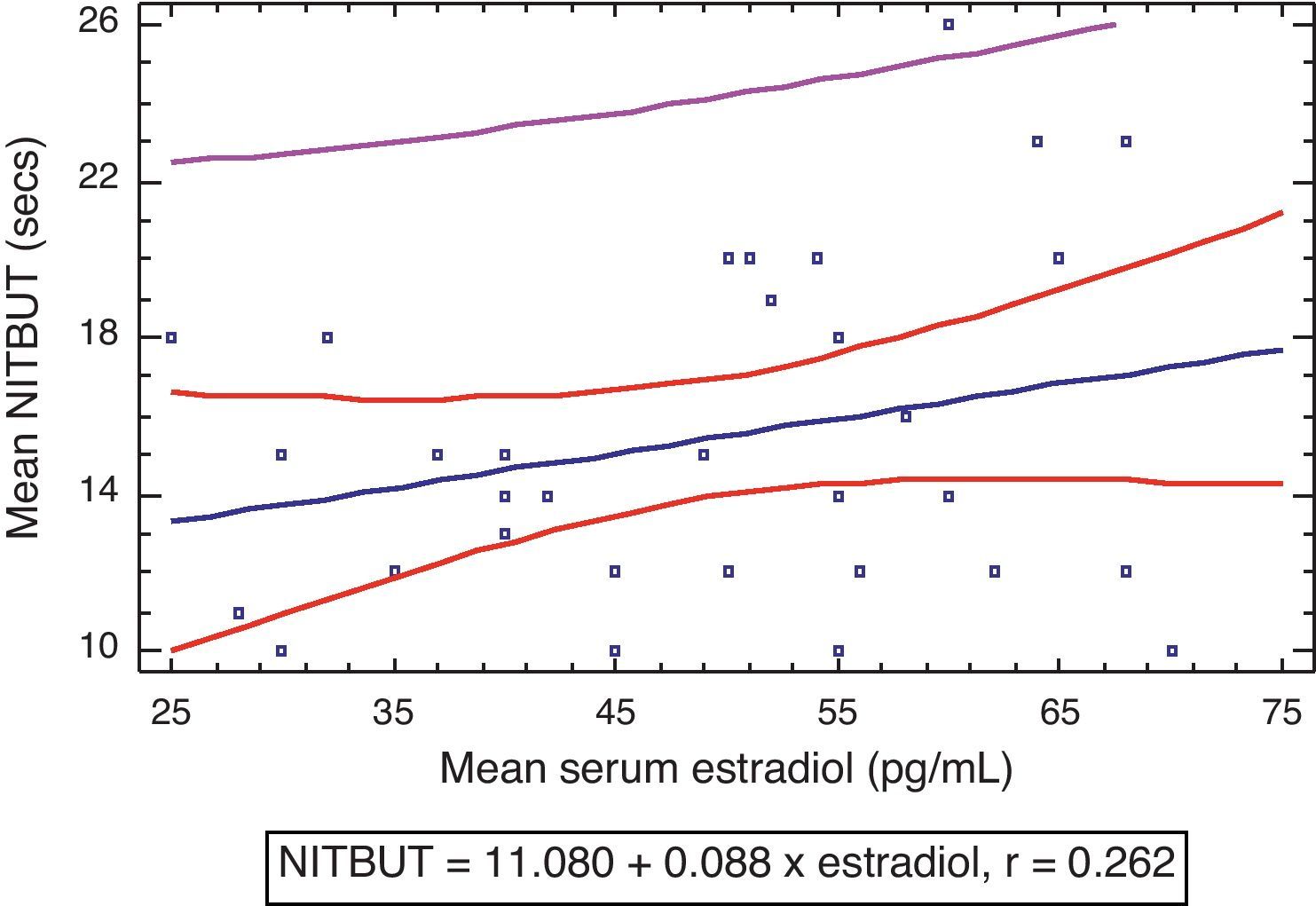
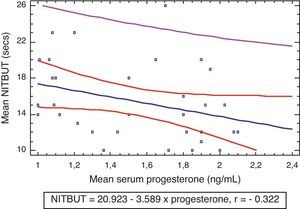
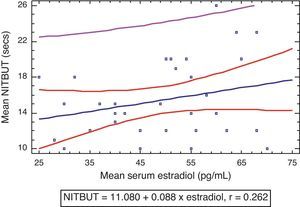
Fig. 3 shows that there was a weak negative correlation between tear stability and serum levels of progesterone in the experimental group (r=−0.322, P>0.05) which was not statistically significant. There was also a non statistically significant weak positive correlation between tear stability and serum levels of estradiol (r=0.262, P>0.05) (Fig. 4).
Tears lubricate, nourish and protect the eyes from dust, irritants and infections. Tears also keep the surface of the eye optically clear and smooth. The imbalances in the composition of tears, may either decrease tear production or encourage excessive tear evaporation. This situation can lead to tear film dysfunction, usually diagnosed as “dry eye”.15
The results of this study revealed that there were no significant differences in the tear secretion (P=0.92) and tear stability (P=0.81) between women on injectable hormonal contraceptives and women not using hormonal contraceptives (P>0.05). The results were consistent with the observations of Tomlinson et al.16 who carried out a similar study on the effects of oral hormonal contraceptives on tear physiology. They16 reported that the oral contraceptives had no effect on normal tear physiology. The observations in this study also agree with that of Kuscu et al.17 who found that HRT had no effect on tear function in post menopausal women. They17 concluded that their observation was the result of the high levels of tear lysozyme and IgA in post menopausal women, using HRT. Taner et al.18 found also that there were no obvious changes in tear film break up time and Schirmer's test values in females on HRT. However, Altintaş et al.19 observed some increase in the tear quality and quantity of women who had been on two years of HRT. The observations of Altintaş et al.19 were different from that of this study.
It was observed that an increase in serum progesterone resulted in slight decrease in tear stability but there were no significant correlations between the serum levels of progesterone and tear stability (r=−0.322, P>0.05). There were also no significant correlations between the serum level of progesterone and tear secretion (r=−0.232, P>0.05).
In overall, injectable hormonal contraceptives had no significant effect on tear secretion and stability on women of child bearing age. It was also observed that there were no obvious correlations between the serum levels of hormones and tear volume and the serum levels of hormones and tear stability. Therefore, this study shows that injectable hormonal contraceptives have no effect on tear production and tear quality in healthy women of child bearing age.
However, further studies may be required to investigate the short term effects of hormonal contraceptives on tear volume and stability of women with dry eyes.
Conflict of interestsThe authors have no conflicts of interest to declare.