To compare changes in the ciliary muscle area at different sectors between pre-presbyopic and presbyopic eyes during accommodation by means of an anterior segment optical coherence tomographer (OCT).
MethodsThe anterior ciliary muscle area was measured in 20 healthy and phakic pre-presbyopic eyes, whose mean age was 23.3±4.4 years, and in 20 healthy and phakic presbyopic eyes, whose mean age was 46.5±5.2 years. The relative change in the cross-sectional area of the ciliary muscle was measured at the nasal, inferior, and temporal sectors between 0 and −3 D of vergence, in −1 D step. A linear model was used to assess the correlation of each eye parameter with the accommodative demand.
ResultsEach population group showed a significant increase in the anterior ciliary muscle area for each sector. The maximum increase in the ciliary muscle area within the pre-presbyopic group was about 30%, and for the presbyopic one was about 25%. At the same time, it was obtained that the larger the vergence, the larger the variability. Furthermore, the linear model showed a positive tendency between the change in the ciliary muscle area of each sector and the vergence for both population groups, which coefficient of determination was in all cases greater than 0.93.
ConclusionThe anterior ciliary muscle area tends to increase with accommodation. The presbyopic nasal, inferior, and temporal ciliary muscle seem to have the same contractile capability as the young presbyopic ciliary muscle. These results might help to increase the evidences in the knowledge regarding the modern understanding of accommodation biometry and biomechanics.
Comparar los cambios en el área del músculo ciliar en diferentes sectores entre ojos con pre-presbicia y presbicia durante la acomodación, por medio de un tomógrafo de coherencia óptica (OCT) de segmento anterior.
MétodosSe midió el área del músculo ciliar en 24 ojos sanos fáquicos y con pre-presbicia, con edad media de 23,3±4,4 años, y en 20 ojos sanos fáquicos y con presbicia, con edad media de 46,5±5,2 años. El cambio relativo en el área transeccional del músculo ciliar se midió en los sectores nasal, inferior y temporal entre 0 y –3D de vergencia, en pasos de –1D. Se utilizó un modelo lineal para valorar la correlación de cada parámetro del ojo con la demanda acomodativa.
ResultadosCada grupo de población reflejó un incremento significativo en el área del músculo ciliar para cada sector. El incremento máximo en el área del músculo ciliar en el grupo de pre-presbicia fue de alrededor del 30%, y para el grupo de presbicia de cerca del 25%. También se observó que cuanto mayor era la vergencia, mayor era la variabilidad. Además, el modelo lineal reflejó una tendencia positiva entre el cambio en el área del músculo ciliar de cada sector y la vergencia para ambos grupos de población, cuyo coeficiente de determinación fue en todos los casos superior a 0,93.
ConclusiónEl área anterior del músculo ciliar tiende a incrementarse con la acomodación. El músculo ciliar en los sectores nasal, inferior y temporal en la presbicia parece tener la misma capacidad contráctil que el músculo ciliar pre-presbicia. Estos resultados podrían ayudar a incrementar las evidencias en el conocimiento relativo a la comprensión moderna de la biometría de acomodación y la biomecánica.
Information about the ciliary muscle has been extracted originally from histological studies in rhesus monkeys because of similarities in the accommodative structures,1 mechanism,2 and presbyopia development relative to timescales.3 This information has been also extracted from human eyes, and studies have pointed out the asymmetry between nasal and temporal sections,4 and its ability to contract through life.5 Nowadays, in vivo and non-invasive visualization of the anterior eye segment are possible due to the advent of high-resolution equipment, such as anterior segment optical coherence tomographers (OCTs) and ultrasound biomicroscopes (UBMs). Some of these equipment allow the study of changes in the anterior eye chamber during accommodation, including the ciliary muscle,6 which is one of the most difficult structures to visualize due to its location.
Previous studies have analysed differences in the ciliary muscle morphology between phakic and pseudophakic eyes by means of its muscle diameter,7 while others assessed changes in the ciliary body during accommodation in children and young adults,8–14 and even the effect of phenylephrine.15 Those studies elucidated differences in the ciliary lens morphology between young and adult eyes, although the muscle's functionality has been shown clearly able to contract under binocular conditions.12 Most of the studies assessed changes on the nasal and temporal ciliary muscle,8,9,16,17 or either on the nasal18 or temporal sectors.10,11,15 Nevertheless, there is no in vivo study assessing multi-sectorial changes in the ciliary muscle during accommodation between pre-presbyopic and presbyopic eyes.
In this scenario, the present study aims to compare changes in the ciliary muscle area at different sectors between pre-presbyopic and presbyopic eyes during accommodation. These results might contribute to increase the evidences in the knowledge regarding the modern understanding of accommodation biometry and biomechanics.
Materials and methodsPatientsThe present study included 40 healthy and phakic subjects who were divided into a pre-presbyopic and presbyopic group. One eye per participant was measured because of the similarity between both eyes in healthy population, and contact lens users were demanded not to wear them at least three hours before the examination session.19 The Donder's method20 was used to ensure that each patient had at least 3 D of subjective amplitude of accommodation monocularly. All measurements were taken during the same session.
The Ethics Committee of the university where the experiment took place gave the ethical approval, and the study was performed in adherence to the tenets of the Declaration of Helsinki. All patients provided written informed consent after the nature and possible consequences of the study were explained to them.
Optical coherence tomographerThe Visante omni (Carl Zeiss AG, Oberkochen, Germany), which is an anterior segment OCT, was used to take all measurements. This system acquires and analyses detailed cross-sectional tomographic images. It obtains measurements of both cornea and anterior segment through the combination of OCT technology and Placido disk topography, and uses an infrared light of 1310nm. The vergence of the fixation target can be adjusted through a set of internal lenses, which was used to evaluate changes in the anterior eye with accommodation. At the same time, these lenses were used to compensate the patient's spheroequivalent.
Experimental procedureA skilled operator acquired all measurements. The ambient lightning conditions were kept stable during the whole procedure. All sets of measurements were taken from 0 D, which was considered as the patient's far point, to −3 D of vergence in −1 D step, and the order of each vergence was selected randomly. Each acquisition was taken 4s after the subject's last blink to achieve a proper tear film spread over the cornea.21 The Raw Image HR mode, which takes 512 A-scans and represents a total area of 10mm in width and 3mm in depth, was used to capture images of the anterior ciliary muscle at nasal, temporal, and inferior sectors. Images on the superior sector were not possible to acquire for all patients because the upper lid blocked the view. In order to improve the image visualization, the OCT prove was directed to the nasal, temporal and inferior sides to capture each ciliary muscle section.
Since the scan was 10mm wide, the OCT-scan was centred on the corneal limbus in all subjects to get the same muscle area in all participants. Furthermore, the examiner that took the manual measurements was masked and not aware of the aims of this study.
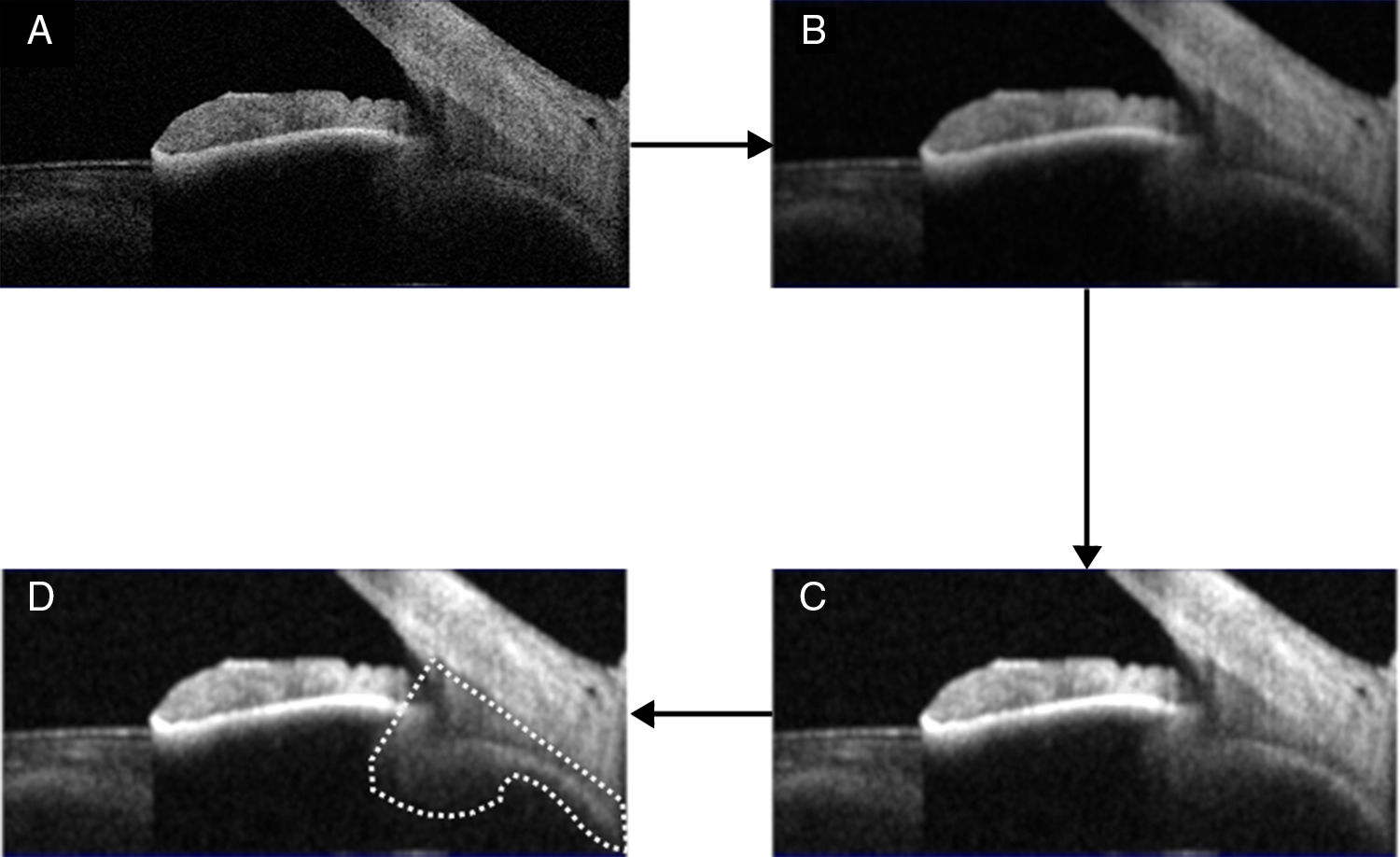
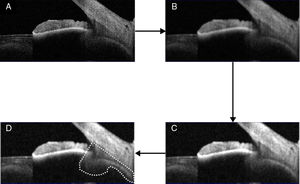
Ciliary muscle analysisThe ciliary muscle of each image was analysed with the ImageJ software, which procedure was displayed in Fig. 1 (panels A–D) as reference purposes. This procedure consisted on converting the original image into an 8-bits one (panel A), including the ciliary processes. Then, a smoothing filter was applied on the image (panel B), and the contrast enhanced with a non-linear histogram equalization (panel C) to highlight different areas and borders and enable the segmentation of the image. After this, the same observer delimited the anterior ciliary muscle area through two lines: One of them was along the border between the sclera and the ciliary muscle, and the other one went through the scleral spur and was perpendicular to the previous line. Then, an automatic selecting tool was applied to select the ciliary muscle area (panel D), in which the area was measured.22 The relative change with respect to the relaxed condition was finally obtained. All measurements were taken in pixels because the ratio of change was reported, and the same experienced and masked observer did all segmentations visually.
Statistical analysisThe results were evaluated using SPSS software v.22 (IBM Corp., Armonk, USA). A repeated measure analysis of variance (rANOVA) was performed to reveal significant differences among changes between vergence conditions within the same sector and population group. The normality of all data sets was evaluated by means of the Shapiro–Wilk test. The ANOVA procedure based on the F statistic is robust under the breach of the normality assumption, provided that the data samples have no important asymmetries or similar distribution shapes.23 Prior to the rANOVA the sphericity assumption was checked using the Mauchly's sphericity test. The Greenhouse–Geisser correction was applied in those cases in which sphericity test was statistically significant.24 The Bonferroni procedure was used as a post hoc test for comparisons between data groups when the rANOVA revealed significant differences between measurements. This method allows for obtaining the significance level for paired differences between the individual conditions. Additionally, a t-test was used to reveal significant differences between both population groups for the same vergence and sector. In all cases, statistical significant differences were considered when the p-value was less than 0.05.
The median values for all changes between vergence conditions were fitted to a linear model using Matlab 2015b (MathWorks Natick, MA, USA) to describe the relationship between the dependent variable (change in the anterior ciliary muscle area for each vergence condition) and the independent variable (each accommodative stimulus). For each regression analysis, the slope and the determination coefficient were obtained. The coefficient of determination (represented as R2) was used to assess how well the regression model fitted the data, which can be interpreted as the proportion of the variance in the dependent variable that is predicted from the independent variable.25
ResultsThe demographic information of the 40 eyes of 40 subjects, who were equally divided between the pre-presbyopic and the presbyopic groups, is included on Table 1. All in all, the mean age in the former group is about 23 years old, and for the latter one is about 47 years old.
Demographic data summary of the subjects included in the study, depending on their age group.
| General (N: 40) | Pre-presbyopic (N: 20) | Presbyopic (N: 20) | |
|---|---|---|---|
| Demographic | 53% Men 47% Women | 60% Men 40% Women | 45% Men 55% Women |
| Age (years) | 34.9±12.7 | 23.3±4.4 | 46.5±5.2 |
| Spheroequivalent (D) | −0.89±2.09 | −1.06±2.46 | −0.73±1.68 |
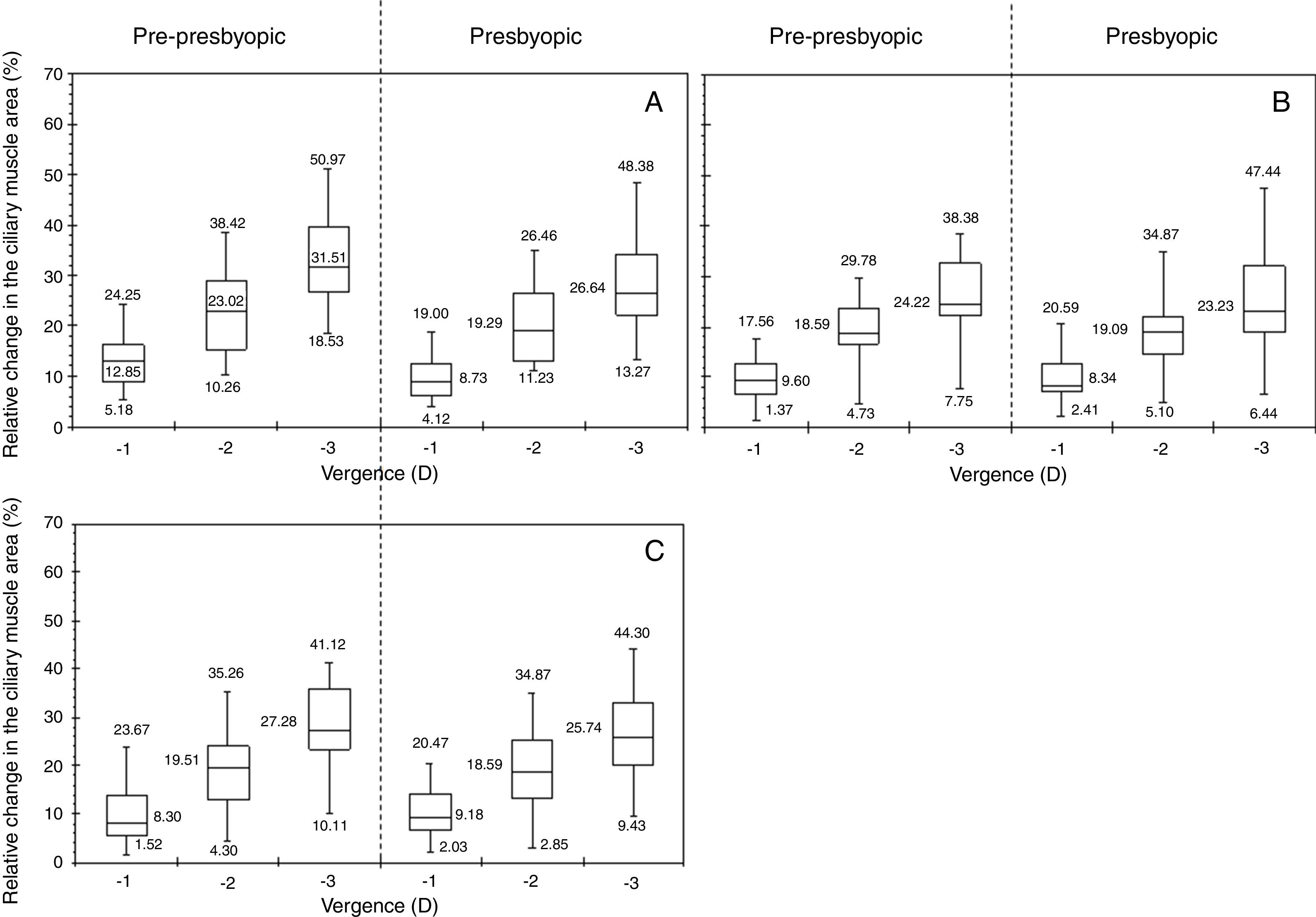
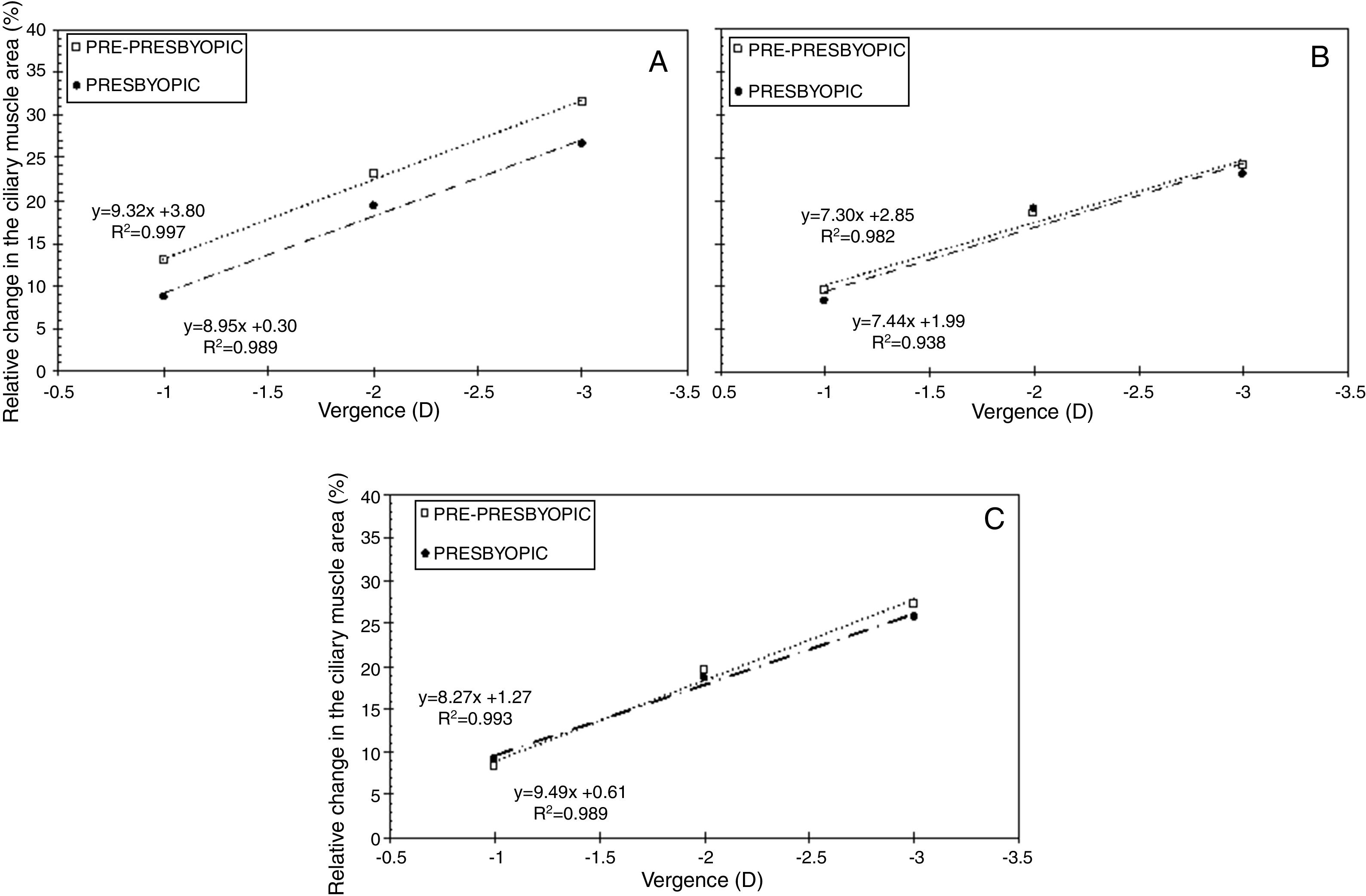
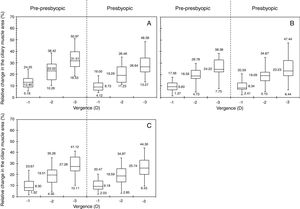
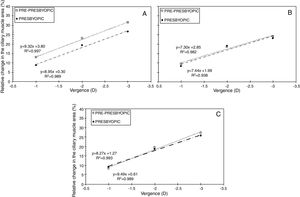
Fig. 2 displays the relative changes in the ciliary muscle area at the nasal (panel A), inferior (panel B) and temporal (panel C) sectors. Fig. 3 includes the linear regression model between the median change in the ciliary muscle area and the vergence for both age groups. For each panel, the dashed line represents the linear fit model for the pre-presbyopic group, and the dashed-dotted line does the linear fit model for the presbyopic one.
Linear regression between the median change in the ciliary muscle area and vergence for the nasal (panel A), inferior (panel B), and temporal (panel C) sector. The dashed line represents the linear fit model for the pre-presbyopic group, and the dashed-dotted line does the linear fit for the presbyopic one.
All in all, both population groups showed an increase in the ciliary muscle area with the vergence for the three sectors measured, and the differences among the vergences were statistically significant (p<0.001) for the same sector and population group. For the pre-presbyopic group, the Nasal sector showed the maximum median increase in the ciliary muscle area, where the median changes were about 12%, 23%, and 31% for 1, 2, and 3 D of accommodation, respectively. The other two sectors showed similar median changes in the ciliary muscle area for the same vergence. Similarly, the linear regressions showed the same tendency, where the Nasal sector resulted with the steepest tendency (Fig. 3).
The presbyopic group showed similar changes in the ciliary muscle area for the three sectors, where the median change was about 8%, 20%, and 24% for 1, 2, and 3 D of accommodation, respectively. Similarly, the linear model fitted resulted with similar slopes for the three sectors analysed. Finally, both groups showed that the smaller the accommodative demand, the smaller the variability.
Inter-group changesThe median change in the ciliary muscle area was similar between both population groups for the same vergence and ciliary muscle sector, in which the differences were not statistically significant (p>0.05). Concretely, the median difference between groups was, at the most, about 5%, where the nasal sector resulted with the largest differences between groups (Fig. 2, panel A). As was expected, the linear correlations showed a positive tendency between the ciliary muscle area and the vergence, in which the slopes between both population groups was similar for the same muscle sector (Fig. 3).
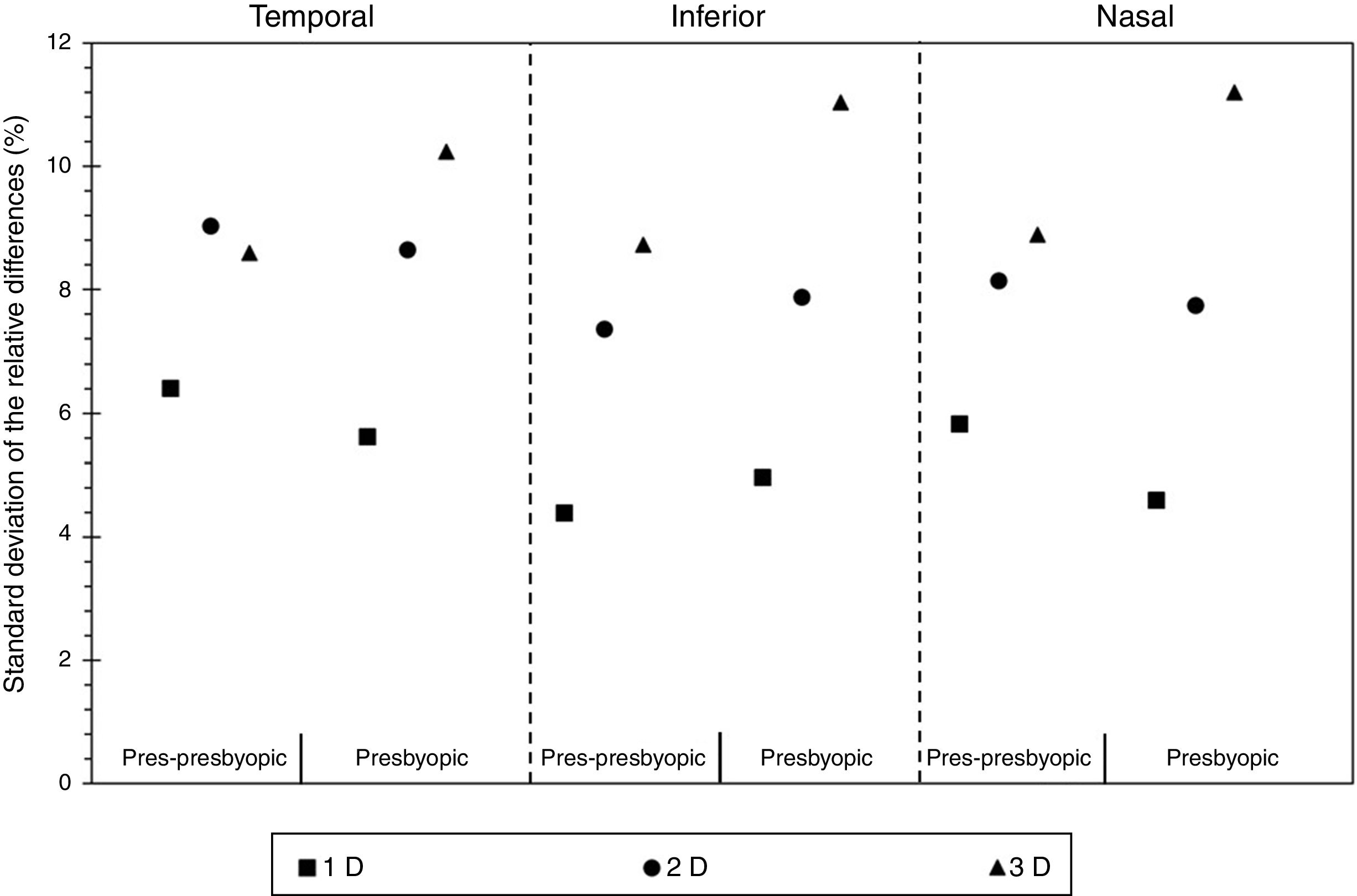
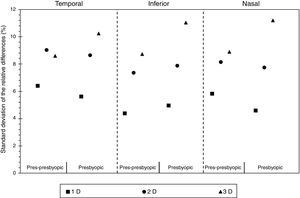
Fig. 4 displays the standard deviation of the relative changes in the cross sectional area of the anterior ciliary muscle as a function of the population group, for each accommodative demand and ciliary muscle sector. The pre-presbyopic group showed smaller variability than the presbyopic group for each sector, especially for big accommodative efforts. Similar results were obtained between both groups for smaller accommodative demands.
DiscussionThe aim of the present study was to compare changes in the ciliary muscle area at different sectors between pre-presbyopic and presbyopic eyes during accommodation, and correlate them with the vergence.
According to the results obtained in the present study, the ciliary muscle area of all sectors tended to increase with the vergence for each population group (panel A, Figs. 2–4). The results showed that the maximum median ciliary muscle increase at the maximum vergence was about 28% for the pre-presbyopic group, and about 25% for the presbyopic one. At the same time, the linear regression (panel B, Figs. 2–4) showed a positive tendency between the change in the ciliary muscle area and the vergence for all sectors and both population groups. This is, the larger the accommodative demand, the larger the change in the ciliary muscle area in pre-presbyopic and presbyopic subjects.
The results obtained in the present study are in agreement with those obtained in previous studies, where changes in the ciliary muscle have been assessed as a function of the vergence.8,10,11,26,27 Sheppard and Davies8 assessed the ciliary muscle morphology by means of an anterior segment OCT in a pre-presbyopic population of 50 subjects. Those authors assessed changes in the nasal and temporal ciliary muscle in terms of anterior length and width at 25%, 50% and 75% of the total muscle length posterior to the scleral spur. The results obtained showed that the anterior portion of the ciliary muscle thickened with accommodation. Similarly, Lewis et al.10 assessed changes in the ciliary muscle morphology as a function of the vergence in 30 children. Concretely, these authors described those changes in terms of the muscle thickness at 1, 2, and 3mm behind the scleral spur, and also at the maximum point. In that study, Lewis and co-workers used an anterior segment OCT to measure changes in the temporal ciliary muscle at three accommodative stimuli (0, 4, and 6 D). The results obtained in that study showed that the temporal ciliary muscle thickened at 1mm and at the maximum, meanwhile the opposite behaviour was observed at 3mm. Another study used an anterior segment OCT to develop a measurement protocol to assess changes in the shape and size of the temporal ciliary muscle during accommodation.11 In that study, 25 adults were included, and their ciliary muscle thickness was measured at the maximum point, and at 1, 2, and 3mm posteriorly the scleral spur Those authors assessed changes in the temporal ciliary muscle at distance vision and at a vergence of −4 D. According to the results obtained in that study, the temporal ciliary muscle area increased at the maximum point and also at 1mm behind the scleral spur. Nevertheless, the ciliary muscle thinned at 3mm from the scleral spur.
Similarly, Richdale et al.26 calculated in vivo changes in the ciliary muscle dimensions as a function of the vergence in emmetropic adults aged between 30 and 50 years. Concretely, the ciliary muscle ring diameter and ciliary muscle thickness were measured with a magnetic resonance imaging (MRI), and an anterior segment OCT, respectively. According to the results obtained, the anterior ciliary muscle thickened with the vergence; meanwhile the posterior thickness and ciliary muscle ring diameter decreased. More recently, those authors27 assessed in vivo changes in the ocular dimensions as a function of the age, refractive error, and vergence in 95 adults aged between 30 and 50 years old. To assess changes in the ciliary muscle with the accommodative demand, the authors used the ciliary muscle ring diameter obtained in a MRI scan, and the ciliary muscle thickness at 1, 2, and 3mm posterior to the scleral spur from an anterior OCT scan. According to the results obtained in that study, the posterior ciliary muscle thinned with accommodation, meanwhile there was no significant change in the anterior ciliary muscle.
According to the results obtained in the present study, the larger the accommodative demand, the larger the dispersion in the ciliary muscle area. From a physiological point of view, these could be due to the effect of microfluctuations,28 which describes that the accommodation fluctuates over a range of about ±0.5 D from the focus point,29 and it has been described to increase with the accommodative demand. Nonetheless, it should be taken into account that there are slightly inconsistent results about the greatest level of fluctuations found either at the near point or at intermediate distances.28 At the same time, the objective amplitude of many subjects in the presbyopic group could be much lower than the subjective amplitude, which was confirmed to be >3 D. As a result, they might not try to exert the same accommodative effort at the highest accommodative stimulus. In fact, the results showed in Fig. 4 in the Presbyopic group showed the highest intersubject variation for the 3 D.
The median change in the ciliary muscle area was similar between the pre-presbyopic and presbyopic groups for the same sector and vergence (panel A in Figs. 2–4), in which no statistical significant differences were obtained between them. At the same time, the regression analyses showed almost the same slope between both population groups within the same ciliary muscle sector (panel B in Figs. 2–4). Thus, from these results can be concluded that the presbyopic ciliary muscle has the same contractile capabilities as the young presbyopic ciliary muscle. Additionally, these results agree with previous studies assessing changes in the ciliary muscle as a function of age.5,8,12
Sheppard and Davies8 assessed in vivo the effect of ageing in human ciliary muscle morphology and contractility during accommodation in 79 subjects whose age ranged between 19 and 70 years. Those authors used high-resolution images by means of an anterior segment OCT at the nasal and temporal ciliary muscle for the patient's far vision and at −4 and −8 D of vergence. From the results obtained, the authors concluded that the human ciliary muscle undergoes age-dependent changes in the morphology that could suggest an antero-inwards displacement of the muscle mass. Nevertheless, those morphologic changes appeared not to affect the ability of the muscle to contract during accommodation, even in established presbyopes. Similarly, another study assessed the ciliary muscle functionality in elder eyes.12 Those authors used their own developed method based on high-speed recording of lens wobbling in order to study the ciliary muscle activity in a small group of pseudophakic eyes. According to the authors, there was a significant activity of the muscle, which was clearly able to contract under binocular stimulation of accommodation.
The present study assessed changes in the ciliary muscle area at the nasal, temporal, and inferior sectors. The superior ciliary muscle was not imaged in all patients because the upper lid blocked the view. The vergence ranges were chosen from the patient's far vision point to −3 D of vergence in −1 D step. Nevertheless, further studies could increase the vergence range. Since there is no easily identifiable landmark in ciliary muscle images to assist in the alignment, the observer did his best to align the scan beam with the midpoint of the pupil. The present study only assessed changes in the anterior ciliary muscle due to the small field of view of the anterior segment OCT used. The image quality of the ciliary body was reduced because the OCT probe was directed to the side of the eye, and the incidental angle could not be ensured to be perpendicular to the surface. The ability of AS-OCT to achieve high quality and reproducible images through opaque media is limited, and the contrast and field of view of the images are limited. Consequently, the nasal-temporal and superior-inferior angles are not possible to image simultaneously. Finally, only 20 eyes per group were included, and further studies should increase the number of eyes per group to obtain more robust conclusions.
To sum up, anterior ciliary muscle area tends to increase with accommodation, although the presbyopic nasal, inferior, and temporal ciliary muscle seem to have the same contractile capability as the young presbyopic ciliary muscle.
Conflicts of interestThe authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
Authors acknowledge financial support from the Ministerio de Economía y Competitivad (Research Project SAF2013-44510-R with ERDF Funds from the European Union), a University of Valencia research scholarship (UV-INV-PREDOC13-110412) awarded to Alberto Domínguez-Vicent, and a Ministerio de Educación, Cultura y Deporte research scholarship (FPU13/05332) awarded to Daniel Monsálvez-Romín.