After a childhood episode of chicken pox, a Varicella-Zoster infection, the viral DNA reside in a dormant state in the dorsal root ganglia. The viruses get reactivated when the individual is immuno-compromised, in adulthood to cause characteristic lesions of Herpes zoster on the skin and the eyes. This case reports a 32 year old female who presented with neuralgia and clinical features of crusting skin ulcers involving the ophthalmic division of the trigeminal nerve, corneal dendritis and anterior uveitis with circumlimbal injection of the right eye. Visual acuity was OD: 6/60 and 0S:6/9. These clinical signs and symptoms were consistent with Herpes Zoster Ophthalmicus (HZO). Further medical laboratory tests showed positive for HIV and patient had a CD4+ count of 350 cells/μl of blood with a viral load of 100,000 copies/μl. Patient was subsequently treated of the Herpes zoster infection with Acyclovir (800mg) prescribed five times daily for 7 days. While, at the HIV/AIDS project facility she was placed on Hyper Active Antiretroviral therapy (HAART): Stavudine (30mg) bid for 2/12, Zidovudine (300mg) bid for 2/12 and Efavirenz (600mg) nocte for 2/12. There was complete resolution of the keratopathy, the visual acuity of OD improved to 6/12 by the 2nd month and the patient was without the experience of post herpetic neuralgia. At present (after 3 months) her CD4+ has increased to 1000 cells/μl. HIV infection should always be considered in patients younger than 65 years with Herpes zoster ophthalmicus.
Después de un episodio de varicela en la infancia, una infección del virus varicelazóster, el ADN vírico permanece en estado latente en los ganglios de la raíz dorsal. Los virus se reactivan cuando el sujeto está inmunodeprimido; en la edad adulta causa lesiones características del herpes zóster en la piel y en los ojos. Se reporta el caso de una mujer de 32 años con neuralgia y características clínicas de úlceras en la piel con costras implicando la división oftálmica del nervio trigémino, dendritis corneal y uveítis anterior con inyección circunlimbal del ojo derecho. La agudeza visual fue de OD 6/60 y OI 6/9. Estos signos y síntomas clínicos eran consistentes con manifestaciones del herpes zóster oftálmico (HZO). Los análisis clínicos posteriores dieron resultado de VIH positivo y la paciente presentó un recuento CD4+ de 350 células/μl en sangre con una carga vírica de 100.000 copias/μl. En consecuencia, la paciente recibió tratamiento con aciclovir (800mg) 5 veces al día durante 7 días para la infección del herpes zóster. Asimismo, en el centro del proyecto HIV/AIDS la paciente recibió tratamiento antirretrovírico de gran actividad (HAART): estavudina (30mg) 2 veces al día, zidovudina (300mg) 2 veces al día y efavirenz (600mg) nocte 2 veces al día. La queratopatía se resolvió completamente, la agudeza visual del OD mejoró hasta 6/12 en el 2.° mes y la paciente no presentó neuralgia posherpética. Actualmente (3 meses después), su CD4+ ha aumentado hasta 1000 células/μl. La posibilidad de infección por VIH siempre debe considerarse en pacientes menores de 65 años con herpes zóster oftálmico.
Chicken pox (varicella) is a highly contagious skin disease primarily in children 2 to 7 years of age.1–3 The causative agent is the Varicella-Zoster virus, a member of the family Herpesviridae, which is acquired by droplet inhalation into the respiratory system.1 Following an incubation period from 10 to 23 days,4 small vesicles erupt on the face or upper trunk. These get filled with pus, get broken and become covered by crusting with an intense sensation of itching. Individuals who recover from chicken pox are subsequently immune to this disease; however they are not free of the virus as viral DNA resides in a dormant (latent) state in the dorsal root ganglia.5
Later in life when this primary infected person is immuno-compromised, the virus may become activated.3,6,7 The viruses migrate along afferent sensory nerves to the skin and eye where they replicate and cause the characteristic lesions.7,8 This reactivated form of chicken pox (Varicella) is termed Herpes Zoster. This report describes a HIV−2 positive patient who presented with Herpes zoster infection which affected the ophthalmic branch of the right trigeminal nerve.
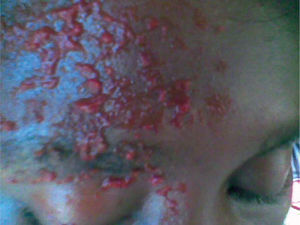
Case reportA 32 year old female presented to the optometric facility of Central ophthalmic clinic in Eket, with complains of burning sensation and deep lancing pain on the surface of the right eye, upper lid, anterior scalp and forehead which followed soon after few days of fever, malaise and headache. Inspection showed skin lesions on the right half of the face, the frontal and the nasociliary i.e. the ophthalmic branch of the trigeminal nerve which have formed crusting ulcers.
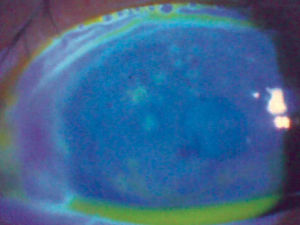
The ocular signs were microdendritic ulcers on the right eye which were mostly peripheral with few central ones that abutted the visual axis. The microdendrites stained with Rose − Bengal. Another ocular sign on the OD was the presence of anterior uveitis with a circumlimbal injection. There was transient diplopia with restricted ocular movement when gaze was directed laterally and upward due to the involvement of the 3rd cranial nerve. Funduscopic examination revealed no retinal involvement. There was no abnormality detected (NAD) on the left eye. Visual acuity was OD: 6/60 and OS: 6/9. In addition the patient disclosed a previous childhood history of chickenpox during her primary school days, about 25 years ago. These signs and symptoms were consistent with Zoster.
Patient claimed to have enjoyed a general good health till this event, though agreed to have recently lost some weight. The patient was referred to the state owned Immanuel General Hospital, Eket for further medical laboratory tests. PCR test for Varicella − Zoster was positive. Genie II rapid HIV screening test was positive for HIV-2. The CD4+ count was 350 cells/μl blood and viral load was 100,000 copies/ml.
In the optometric facility, patient was treated for Herpes Zoster Ophthalmicus (HZO) with oral Acyclovir (800mg) prescribed 5 times daily for 7 days and topical prednisolone acetate (1%) 2 drops instilled four times daily for 7 days. The pupil of the right eye was kept dilated with Atropine (1%) of which 1drop was instilled three times daily for one week.
After one month, on appointment she returned to Immanuel General Hospital, to the Global HIV/AIDS initiative of Nigeria (GHAIN) project facility for her counseling. She was categorized “stage 2B” by the CDC clinical and immunological staging14 and placed on Hyper Active Antiretroviral Therapy (HAART): Stavudine (30mg) bid × 2/12, Zidovudine (300mg) bid × 2/12 and Efavirenz (600mg) nocte × 2/12.
After 3 months on her HAART, her CD4+ count was increased to 1000 cells/μl and the viral load markedly reduced to less than 1,000 copies/ml.
Her ophthalmic and Acyclovir medications prevented synaechia and the chronic form of cornea ulcer. There was a total resolution of the keratopathy. The visual acuity (VA) on the right eye improved to 6/12 by the second month; and presently the VA stands at 6/6+ on either eye. The patient also restored is a smooth ocular motility without the experience of diplopia. Patient has been also without the experience of a post-herpetic neuralgia.
DiscussionA Herpesviridae is a double stranded DNA virus, with the outstanding property of establishing a lifelong persistent infection in their hosts by undergoing reactivation; with the reactivated infection being clinically quite different from the disease caused by primary infection.3
Varicella (Chickenpox) and Herpes zoster (Zoster) are different conditions caused by the same herpesviridae called Varicella−Zoster. While Varicella is common in children, Herpes Zoster mainly affects the elderlies.3,8,9 Throughout the world the incidence rate of herpes zoster every year ranges from 1.2 to 3.4 cases per 100 healthy individuals, increasing to 3.9–11.8 per 1,000 individuals among those older than 65 years.8
The current theory of etiology is that after an attack of chicken pox and its attendant viraemia, some virus spread along the sensory nerves from the skin lesions to be retained in the ganglia adjacent to the spinal cord (dorsal root ganglion) or the semilunar ganglion in the base of the skull.7,9 Later, under the influence of trigger factors particularly reduced immunity by such factors as psychological, physiological stress, age or AIDS, the virus reactivates and migrates along afferent sensory nerves to the skin and eye where it causes the characteristic facial lesions.2–7 This is directly the result of viral infection, antigen-antibody reactions, delayed cell mediated hypersensitivity reactions, denervation and ischaemia induced by vasculitis.
Approximately 15% of all cases of Herpes zoster affect the frontal, lacrimal and nasociliary branches− that is the ophthalmic division of the trigeminal nerve.9 The condition is then referred to as Herpes Zoster ophthalmicus (HZO). The HIV positive population have a 15–25 times greater prevalence of zoster compared to the general population10 and the risk of developing HZO has been reported to be 6.6 times higher than that of the general population.11
As occurs with other opportunistic infections in HIV, Herpes zoster can be more debilitating with greater skin and ocular involvements. HZO is usually described by a prodrome of fever, malaise and headache which typically herald the neuralgia component which precedes the cutaneous lesion of HZO. The neuralgia varies from itching, burning sensation to severe deep boring or lancing pain which is either persistent or intermittent. These may totally resolve within the first 4 weeks. Trigeminal neuralgia is caused by demyelination of trigeminal sensory fibres resulting from compression by an overlying blood vessel or infiltrations.
The skin lesions of Herpes zoster are histopathologically identical to those of Varicella.3 The rashes may involve one or all the three branches of the ophthalmic distribution of the trigeminal nerve.9 Initially the lesion is maculopapular and then becomes pustular. The pustules subsequently burst to form crusting ulcers (figure 1). The involvement of the nasociliary nerve (Hutchinson's sign) which supplies the side of the nose correlates significantly with the subsequent development of ocular complications.12
Conjunctivitis is common and always associated with vesicles on the lid margins. This usually resolves within one week. If chronic, a mucus-secreting conjunctivitis may be accompanied with lipid-filled granulomata under the tarsal.
Episcleritis occurs in about 1/3 of cases at the onset of the rash but it is usually concealed by the overlying conjunctivitis; scleritis is much less common. When present, scleritis frequently becomes chronic and gives scleral atrophy, while a neglected sclerokeratitis yields a progressive scarring of the cornea.
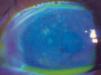
Corneal complications occur in approximately 65% of cases with HZO. Punctate epithelial Keratitis develops in about 50% of patients within 2 days of the onset of the rash.10 The foci consist of swollen epithelial cells containing replicating virus. The lesions stain with Rose Bengal and are usually peripheral, multiple, raised, small and occasionally coated with mucous.
Microdendritic ulcers are also another mark of epithelial disease and appear within 4–6 days. HZO microdendrites are common and have no central ulceration or “terminal bulbs” like dendritic ulcers. The ulcers are usually peripheral, broader, more plaque like and more stellate shape than dendritic (figure 2).
Nummular keratitis is seen beneath the Bowman's membrane in 33% of patients, 10 days after the onset of rashes. The lesion is characterized by multiple fine granular deposits which are surrounded by a halo of stromal haze. Lesions may resolve without trace or become indolent with chronic cellular and lipid infiltration that impair vision.
Disciform keratitis most at times follows nummular keratitis. It appears in 5% of cases 3 weeks after the onset of rashes and usually central in location. If neglected there will be scarring, vascularization and deposition of lipid.
Mucous plaque keratitis develops in about 5% of cases at any time from two weeks of onset rashes. It is marked with a sudden onset of ciliary injection and mucous plaque deposits on the surface of a diffusely swollen corneal epithelium. The white-grey plaque is adherent to the surface epithelium, with sharply demarcated margins and may be branched or linear. The plaques disappear after 3 months leaving a faint diffused corneal haze.
The neurological complications are cranial nerve palsies particularly of the 3rd cranial pair. Recovery occurs spontaneously within six months. Optic neuritis occurs in about 1 in 400 cases.
Many cases of acute retinal necrosis associated with Varicella-Zoster virus are without other clinical features of varicella or zoster infection elsewhere.9 Hence a laboratory diagnosis for VZV is recommended in acute retinal necrosis when there are no other ocular complication or skin lesions.
A rise is specific antibody titer can be detected in the patient's serum by tests like Complement Fixation, Neutralization Test (in cell culture), Indirect Immunofluorescence and Enzyme immunoassay.
When vesicles are present, diagnoses in the laboratory can also be made by staining smears of scrapings or swabs of the base of vesicles and multinucleated giant cells are seen. These are absent in non-herpetic vesicles.3 Immunoflourescein staining of smear can be used as well to detect intra cellular antigens.
In rapid diagnosis, in electron microscope examination, Varicella-zoster virus − specific antigens or viral DNA can be detected in vesicle fluid, in extracts of crust or in biopsy material.
The virus can also be isolated from vesicle fluid using cultures of human cell in 3–7 days. This is inoculated promptly since the virus is very labile in vesicle fluid.
The presence of HZV in a young otherwise healthy patient should provide pause for the consideration of underlying HIV.
The laboratory diagnosis for HIV can be by viral isolation and culture, or by using assays for viral reserve transcriptase activity or viral antigens. However, diagnosis is best through the detection of specific anti HIV antibodies in the blood. These antibodies may be detected by GENIE or HIV AG (Abbott) rapid tests, the SUDS (Murex) 10 minute − assay, Enzyme-linked Immunoabsorbent Assay (ELISA), Indirect immunofluorescence, Immunoblot (Western Blot) and radioimmuno precipitation methods.
HIV destroys the immune system for the fact that the virus-specific protein glycoprotein 120 envelope protein binds to the CD4 glycoprotein plasma membrane receptor on CD4+ T-cells, and HIV accumulates in lymphoid organs in large quantities even before symptoms appear. Thereafter the lymph nodes internal structure collapses due to viral replication. This leads to a decline in the number of lymphocytes within the lymph nodes and results in a selective depletion of the CD4+ T-cell subset.13 When this CD4+ population declines, it leaves the infected person open to opportunistic infections.1,13 In 1993 the CDC defined AIDS to include all HIV infected persons who have fewer than 200 CD4+ cells per μl or a CD4+ cell percentage of total lymphocytes of less than 14. Healthy individuals have about 1000 of such cells in every μl of blood. In HIV infected individuals, the number declines by an average of about 40 to 80 cells per μl each year.13 When the CD4+ cell count falls to between 400 and 200 per μl the first opportunistic infections and disease processes usually appear.14
Herpers zoster may occur at any stage of HIV infection and be its first clinical presentation.15,16 The risk of developing herpes zoster is not associated with duration of HIV infection or rapid progress to AIDS, nevertheless patients with CD4+ counts between 50 and 200 cells/μl seem to be at the highest risk of a herpes zoster event.17
At present there is no complete cure for AIDS. Primary treatment is directed at reducing the viral load and increasing the CD4+ count as well as at treating the opportunistic infections.
Treatment of Herpes Zoster Ophthalmicus is with oral antiviral drugs, mydriatics and topical corticosteroids. Early treatment with Acyclovir 800mg 5 times a day; Famicidovir 500mg or Valacylovir 1 gram t.i.d for 7 days reduce ocular complications. Patients with keratitis or uvetis require topical corticosteroids e.g. prednisolone acetate 1% instilled q.i.d initially, lengthening the interval as symptoms improve. The pupil should be dilated with Atropine 1% or scopolamine 0.25% 1 drop t.i.d., intraocular pressure must be monitored and treated if it rises significantly above normal values. For the symptoms, the use of a brief course of high doses of corticosteroids to prevent post herpetic neuralgia in patients more than 60 years who are in good general health remains controversial.12
In our optometric facility this case was treated for Herpes Zoster Ophthalmicus (HZO) with oral Acyclovir (800mg) prescribed 5 times daily for 7 days and topical prednisolone acetate (1%) 2 drops instilled four times daily for 7 days. The pupil of the right eye was kept dilated with atropine (1%) of which 1 drop was instilled three times daily for one week. There was a total resolution of the keratopathy. The visual acuity (VA) on the right eye improved to 6/12 by the second month; and presently the VA stands at 6/6+ on either eye. The patient also restored a smooth ocular motility without the experience of diplopia. Patient has been also without the experience of a post-herpetic neuralgia. A post-herpetic neuralgia affects about 7% of patient and may lead to depression.9
The antivirals currently approved for use in HIV disease to increase the CD4+ count and reduce the viral load are of three types.18
- 1.
Nucleoside reverse Transcriptase inhibitors (NRTIS). Examples are Zidovudine (AZT), Stavudine (d4t), Lamivudine (3TC), Didanosine (ddi), Abacavir (ABC), Zalcitabine (ddc) and Emtricitabine.
- 2.
The Nonnucleoside Inhibitors of Revene Trans criptase (NNRTIS). Examples are Neviropine (NVP), Efavirenz (EFV) and Delaviridine (DLV).
- 3.
The protease inhibitors (PI) work by blocking the activity of the HIV protease and thus interfere with vision assembly. Examples are Saquinavir (SQV) indinavir (IDV), Ritonavir (RTV), Nelfinavir (NFV), Amprenavir (APV) and Lopinavir (LPV).
Therapy with combinations of antiretroviral, such as the combination of AZI, ZTC and RTV, at high dosages to prevent the development of resistance is referred to as Highly Active Antiretroviral Therapy (HAART). It suppress viral replication to below limits of detection in plasma and decrease viral loads in lymphoid tissue, allowing the recovery of immune response to opportunistic pathogens.19,20
The patient was placed on Hyper Active Antiretroviral Therapy (HAART): Stavudine (30mg) bid × 2/12, Zidovudine (300mg) bid × 2/12 and Efavirenz (600mg) nocte × 2/12. After 3 months on her HAART the viral load was markedly reduced to less than 1,000 copies/ml and her CD4+ count increased to 1000 cell/μl.
If HIV treatment is started early enough the immune system (as monitored by the increased CD4+ count) will be recovered after HIV levels have fallen.21 This prevents the development of opportunistic infections. Hence the need for early detection of HIV. Recognition of potential opportunistic infections in selected patients will help achieve the goal of early detection of HIV.
HIV infection should always be considered in patients younger than 65 years with herpes zoster ophthalmicus.15,22
Conflict of interestsNo conflict of interests.