The use of swimming goggles (SG) has demonstrated to alter different ocular parameters, however, the impact of wearing SG on the tear film stability remain unknown. The main objective of this study was to determine the short-term effects of wearing SG on tear film surface quality break-up time (TFSQ-BUT).
MethodsTwenty-eight young healthy adults (14 men and 14 women) wore a drilled SG, and TFSQ-BUT was measured before, during and after SG use. Dynamic-area high-speed videokeratoscopy was used for the non-invasive assessment of TFSQ-BUT.
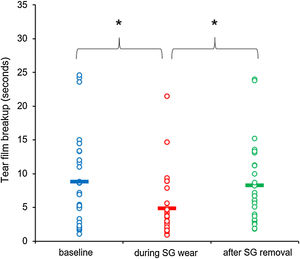
ResultsTFSQ-BUT was significantly reduced while SG wear in comparison to the baseline measurement (4.8 ± 4.5 s vs. 8.8 ± 6.9 s; corrected p-value = 0.017, d = 0.57, mean difference = 4.0 [0.6, 7.3]; 45% reduction). Immediately after SG removal, TFSQ-BUT rapidly recovered baseline levels (8.2 ± 5.9 s vs. 8.8 ± 6.9 s; corrected p-value = 0.744). The impact of wearing SG on TFSQ-BUT were independent of the gender of the participants (p = 0.934).
ConclusionThe use of SG induces a TFSQ-BUT reduction, with these changes returning to baseline levels immediately after SG removal. These data may be of relevance for the management of dry eye patients, who need to avoid circumstances that exacerbate tear film instability. Nevertheless, these results must be interpreted with caution since the experiment did not entirely mimic real-life conditions (e.g., eye cup piece drilled, time of exposure, environmental conditions). Future studies should consider the inclusion of dry eye patients and older individual in order to explore the generalizability of these findings.
Blinking, apart from contributing to the maintenance of ocular surface humidity, favors the tear drainage and spreading of tear lipids across the precorneal film.1 Immediately after a blink, the tear film undergoes formation (build-up), being followed by a phase of relative tear film stability, and finally, the tear film break-ups. Precisely, tear film surface quality break-up time (TFSQ-BUT), which is defined as the interval following a blink to the first occurrence of dry spots on the cornea,2 is a key component in the diagnostic of dry eye disease (DED).3
There is scientific evidence (see a review by Wolkoff4) that different environmental factors (e.g., low relative humidity, high temperature or altitude) meaningfully alter the precorneal tear film. Also, different occupational activities such as visual tasks that lead to insufficient eye blink frequency and increase of the ocular surface area (e.g., prolonged near-work, use of electronic displays or adopting a vertical gaze position [aircraft pilots]), personal factors (e.g., age, gender or medication use) or wearing contact lenses have also been associated with instability of the precorneal tear film, leading to ocular dryness, irritation and discomfort.4,5,14–18,6–13
Due to the increasing frequency of DED, eye care professionals are constantly investigating for the most pertinent strategies for DED treatment.19 Despite the scarce scientific evidence, physical exercise training has been recently proposed as a potentially useful strategy in the management of DED,20,21 since lower levels of physical activity have been associated with DED. 21 In this regard, swimming may be one of the most popular recreational activities advisable for healthy and clinical individuals.22 However, this type of physical activity is not exempt of potential risks, including the eye health, since the chemical compounds used for swimming pool disinfection have demonstrated to be potentially harmful for the ocular surface health,23,24 suggesting that use of swimming goggles (SG) is highly recommended. Additionally, wearing SG has showed to promote acute IOP elevations25,26 and increase the axial length of the eye,27 and thus, eye care specialists discourage its use when avoiding IOP or ocular biometrics changes are desirable. Also, the use of SG has demonstrated to alter the anterior eye biometrics,28 and thus, it is plausible to expect that the changes caused by SG wear in the corneal may negatively affect the tear film stability. From a clinical point of view, a reduced term film stability is associated with contact lens intolerance,29 dry eye symptomatology (i.e., foreign body sensation, dryness, soreness, etc.),30 and ocular hyperemia.31
As indicated above, the measurement of tear break-up time is one of the most used diagnostic tests for DED, which is commonly performed by the instillation of fluorescein. However, the use of fluorescein has showed to alter tear stability, and thus, the validity and repeatability of this measurement is questioned.32–36 Notably, recent advances in objective methods for detecting dry eye have permitted to incorporate non-invasive techniques for the evaluation of tear break-up time, which have demonstrated a considerable potential for the objective detection of dry eye.37–41 Between these techniques, the automated non-invasive measure of tear film surface quality break-up time (TFSQ-BUT) derived from Placido disc videokeratography have showed to be more repeatable than clinician-derived estimates (e.g., slit lamp illumination technique),30 and thus, this examiner-independent method is highly recommended for clinical and research settings.34,42
In view of the scientific literature, despite the SG wear may have some beneficial and detrimental effects on the ocular surface health, its possible impact on tear film stability, which is an important indicator of DED, it has not been sufficiently investigated. In view of the aforementioned limitations, the present study aimed to assess the influence of wearing SG on TFSQ-BUT in healthy young adults, as measured by non-invasive dynamic-area high-speed videokeratoscopy. Complementary, the possible gender-differences on the TFSQ-BUT changes caused by SG wear were tested. Based on previous research, the tension transmitted by the goggle headband on the orbital tissue may alter the tear film, as it has been demonstrated for other ocular parameters.26,27 Regarding gender differences, there are no previous studies that have investigated whether the ocular physiology responses to SG wear differ between men and women, and thus, the lack of previous evidence does not allow us to formulate any hypothesis in this regard.
MethodsParticipantsThis study is the first of its nature, and thus, there were not applicable data for sample size calculation. An a priori power analysis (GPower 3.1 software43), considering an assumed power of 80, alpha of 0.05 and effect size of 0.25, was conducted, which projected a required sample size of 28 (14 participants per group). Based on this, twenty-eight young healthy adults (14 women) took part in the present study. All participants accomplished the following inclusion criteria: (a) had no history of ocular surgery or orthokeratology, (b) be free of any systemic or ocular disease, (c) not be taking any ocular or systemic medication, (d) not use artificial tears, lid hygiene products or warm compresses twelve hours before the experimental session, and (e) having a score ≤12 on the Ocular Surface Disease Index (OSDI).44 Additionally, contact lens wearers (five participants were users of soft contact lenses with a water content higher than 55%) were asked to avoid the use of their contact lenses for 8 h prior to attending the experimentation in order to avoid the influence the short-term contact lens wear on the tear film and corneal surface.45 This study was conducted in conformity with the Declaration of Helsinki, and permission was provided by the University Institutional Review Board (438 / CEIH / 2017).
Experimental designA specific SG model (Nabaiji, Decathlon Group Inc., Villeneuve d'Ascq, France) was used, which consisted of two separated rigid plastic eye cups with a rubber cushioning seal surrounding the lip of each cup and a non-adjustable elastic strap. Vertical and horizontal goggles widths of the cup were 45 mm and 33 mm respectively, from internal rubber seals of each eyepiece. Aiming to assess the TFSQ-BUT during SG wear and based on the experimental design of Paula et al.,26 who investigated the intraocular pressure changes induced by SG wear, the central part of the plastic eye piece of the right eye was drilled, allowing us to obtain a measurement of non-invasive TFSQ-BUT while wearing the SG.
Instruments and measurementsFor the non-invasive assessment of TFSQ-BUT, the CA-800 Corneal Analyser (Topcon, Tokyo, Japan) was employed, using its tear breakup time module. This device uses high-speed videokeratoscopy, which allows to determine the temporal changes in the specular reflection of a grid pattern (Placido discs) projected onto the tear film. High-speed videokeratoscopy permits the analysis of the entire visible corneal surface, and it has demonstrated to be the most precise method for measuring TFSQ-BUT.46,47 In particular, the CA-800 Corneal Analyser records the time needed to break the 5% of the available area in two consecutive photokeratoscopic images (visible corneal surface). The right eye was always chosen for the assessment of TFSQ-BUT, and it was based on the high level of between-eyes correlation (coefficient of determination = 0.94) found for the tear film breakup time in healthy subjects.48 TFSQ-BUT was measured before SG wear (baseline measurement), while wearing the SG for 3 min (during SG wear measurement), and immediately after wearing the SG for 5 min (after SG removal measurement). These points of measurement were chosen based on two recent studies assessing the impact of SG wear on intraocular pressure and anterior eye biometrics, in which participants were asked to wear the SG during 5 min and the ocular parameters were measured while wearing the googles and immediately after removing the SG.26,28
ProcedureParticipants attended two experimental sessions with the first verifying inclusion and exclusion criteria. Data from the second session were submitted to main analyses, and in this session, TFSQ-BUT was measured before SG wear, after 3 min of wearing the SG, and just after SG removal. For data acquisition, participants were asked to place their chin and forehead in the corresponding supports of the CA-800 Corneal Analyser. Before each TFSQ-BUT measurement and following the manufacturer instructions, participants were instructed to voluntary blink before starting data acquisition, and then, to avoid blinking until the TFSQ-BUT measurement was obtained. All, measurements were conducted under constant environmental conditions (∼22 °C and ∼65% humidity).
Statistical analysisA mixed analysis of variance (ANOVA), considering the point of measure (before, during, after) as the only within-participants factor and the participants´ gender (men and women) as the only between-participants factor was carried out for the TFSQ-BUT. The magnitude of the differences was assessed through the Cohen’s d effect size (ES) and the mean difference with the corresponding 95% confidence interval. ESs were interpreted following the recommendations given by Cohen 1998: negligible (<0.2), small (0.2–0.5), moderate (0.5–0.8), and large (≥0.8). For multiple comparisons (ANOVAs), the magnitude of the differences was explored by eta squared (η²). Statistical significance was set at an alpha level of 0.05, and post hoc tests were corrected with Holm–Bonferroni procedure.
ResultsParticipant’s age was 21.8 ± 1.8 years (men = 21.4 ± 2.2 years, and women = 22.1 ± 1.3 years), and OSDI scores were 4.0 ± 3.7 (men = 3.8 ± 3.3, and women = 4.2 ± 4.2). No differences were observed for age and OSDI scores when men and women were compared (p = 0.324 and 0.784, respectively).
A statistically significant effect of wearing SG on TFSQ-BUT (F2, 54 = 4.204, p = 0.020, η² = 0.135) was obtained. Post-hoc analyses evidenced that the TFSQ-BUT was lower during SG wear in comparison to the baseline (corrected p-value = 0.017, d = 0.57, mean difference = 4.0 [0.6, 7.3]) and after SG removal (corrected p-value = 0.035, d = 0.48, mean difference = −3.4 [−6.8, 0.0]) measurements. However, no differences were observed between the baseline and after SG wear measurements (corrected p-value = 0.744) (Fig. 1). For its part, no statistically significant differences were obtained for the main effect of gender and the interaction SG use x gender (F1, 27 = 0.007, p = 0.934; and F2, 54 = 0.255, p = 0.776).
DiscussionThe effects of SG wear on TFSQ-BUT in men and women were assessed. The data revealed that TFSQ-BUT is reduced while wearing SG, with TFSQ-BUT returning to baseline levels immediately after SG removal. The present outcomes converge with similar studies that have evidenced acute effects of SG wear on different ocular indices such as intraocular pressure, corneal thickness, anterior chamber angle and axial eye length,25–28 although these findings should be cautiously interpreted since the experimental conditions did not fully mimic real-life conditions (eye piece of the SG was drilled). Despite evidence that the use of SG prevents the detrimental effects of swimming pool chlorine on the corneal epithelium,23 these results suggest that SG wear may have a negative impact on tear film stability, as assessed by TFSQ-BUT.
Leaving aside the personal characteristics (e.g., age or gender) and lifestyle habits as factors associated with DED,49 the TFSQ-BUT has demonstrated to be altered by multiple causes such as wearing contact lenses, computer work, adverse environmental conditions (e.g., low humidity, air conditioning, pollution), medication use, and refractive or cataract surgery among others.4–18 In addition, these data revealed that SG wear acutely diminishes tear breakup time, suggesting that the use of SG leads to tear film disruption and possible ocular surface damage. Therefore, the use of SG may be also taken into account when considering the circumstances or conditions in which the tear film stability could be deteriorated.
Previous researchers have argued that the impact of SG wear on different ocular parameters is due to the tension transmitted by the goggle headband which compresses the orbital tissues and vasculature.25,27 A recent study has demonstrated that 3.5 min of SG wear are enough to induce a considerable thinning (∼55 µm) of the central corneal thickness,28 with corneal thinning being associated with higher levels of tear osmolarity.50 It has been argued that tear film breakup is the result of a linear thinning of tear film between blinks, which may be due to flow of tears in three directions: outward (i.e., evaporation), inward flow of water into the corneal epithelium and tangential flow along the surface of epithelium.51 Although, there is consensus that evaporation is the main factor associated with tear film thinning.52 the role of the tangential flow mechanism in tear film stability should be taken into account since changes in the corneal morphology has been found while SG wear.28 Based on this, it could be speculated that the reduction in central corneal thickness as consequence of the mechanical pressure elicited by the goggle on the ocular and periocular tissues may be responsible of the impaired tear film stability while wearing SG. A 45% reduction of TFSQ-BUT was found while wearing SG in comparison to the baseline value, with the average value obtained during goggle wear being of approximately 5 s (4.8 ± 4.5 s). Recently, the Asia Dry Eye Society has recommended to consider a cutoff value of less than 5 s for the diagnosis of dry eye,3 and the results obtained in the current study during SG wear are slightly below this cutoff value. It should be noted that there is a considerable variability between the different methods used for the assessment of tear film stability,53 and thus, this result should be cautiously interpreted in this regard.
Evidence suggests that the major causes of an increased instability may be related to the quality of the “tear binding surface” and the efficacy of the eyelids during blinking.54 The compression elicited by the SG on the eyelids and orbit could affect the blinking pattern, inducing an increased surface tension and an higher evaporation and dewetting.51 However, this fact may be irrelevant in the current study since participants were asked to avoid blinking during TFSQ-BUT assessment. In addition, changes in environmental humidity and temperature have demonstrated to affect tear film stability.14,18 These effects may be insignificant in this study since the plastic eye piece of the SG was drilled, and thus, negligible temperature and humidity variations would be expected. Abusharha et al. (2016) observed that the stability of tear film was impaired at temperatures lower than 10 °C and that the evaporation rate increases with temperatures higher than 20 °C, being 25 °C the temperature limit for a normal tear film.18 Korb and colleagues55 used a modified SG to manipulate the level of periocular humidity and found that eyes exposed to ambient room conditions did not change the thickness of the tear film lipid layer whereas eyes exposed to conditions of high humidity experienced a significant increase of lipid layer thickness. Therefore, humidity and temperature values should be considered when comparing this study with those performed in real-life conditions. Additionally, it is plausible to expect that SG wear could cause a mechanical effect on the ocular structures, which may affect tear production or drainage. This possibility is supported by the rapid recovery of TFSQ-BUT after SG removal, although it needs to be assessed in future investigations. In regard to the differences in tear film stability between men and women, there are some controversial results in the scientific literature. For example, Ozdemir and Temizdemir56 found that tear break up time was similar between men and women, whereas Maïssa and Guillon10 reported that men have a longer non-invasive break up time when compared to women, although these differences were only significant on individuals less than 45 years old. Here, the TFSQ-BUT decreased during SG wear regardless of participant´s gender.
As previously mentioned, the avoidance of environments and situations that have a detrimental effect on tear film stability is paramount to minimize ocular discomfort.4 In this sense, the use of SG is highly desirable in order to avoid the ocular side effects associated with swimming pool chlorine.23,24 However, wearing SG also induces undesirable effects on intraocular pressure and anterior eye biometrics,25–28 as well as on tear film stability, as found in the current study. Eye care specialists should consider these effects of SG wear on the ocular physiology in order to prevent or minimize undesirable ocular side effects. However, there are several aspects that may limit the generalizability of these findings, and they should be acknowledged. First, a specific SG model was used on this study, and thus, the effects of different SG models on tear film stability require further investigation. Second, it should be noted that due to methodological requirements, the anterior section of the eye cup piece was drilled in order to perform an evaluation of TFSQ-BUT during SG wear. Therefore, the present findings need to be cautiously interpreted in this regard, since this experiment did not entirely mimic real-life conditions, in which the negative air pressure effects and the closed space into the SG may have an additional impact on tear film stability. Third, in this study, the effects of SG wear during a short-period of time (5 min) on TFSQ-BUT were tested, however, wearing SG for longer periods of time may lead to different results. Fourth, our study was carried out in a laboratory setting, and SG are used in water and in specific humidity and temperature conditions, which may also influence tear film stability. Therefore, the current results must be carefully interpreted in this regard. Also, the long-term effects of using SG (habitual swimmers) on the tear film should be address in future investigations. Remarkably, the consideration of other parameters related to tear film production or evaporation, as well as those corresponding to specific tear film layers (e.g., lipid) may incorporate novel insights into the impact of SG on the tear film quality. Lastly, there is evidence that older individuals and dry eye patients have a lower tear breakup time in comparison to controls,3,10,56 and the practice of swimming is highly recommended for older and clinical populations, since its impact on the musculoskeletal system is low and the incidence of injury is rare.22 Moreover, the assessment of the impact of SG wear on tear film quality on these populations deserves further investigation.
ConclusionThe present findings show that wearing SG induces a tear film breakup time reduction, with these values returning to baselines levels just after goggle removal. This result may have important implications for the ocular surface health, being of special relevance in DED patients. Due to technical requirements, the anterior section of the eye cup piece was drilled in order to assess tear film stability during SG wear, and thus, the experimental design did not entirely mimic real-life conditions. The inclusion of DED patients and older individuals is needed to corroborate the external validity of the present outcomes.
Conflict of interestAll authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest or non-financial interest in the subject matter or materials discussed in this manuscript.