Corneal refractive surgery procedures are widely performed to permanently correct refractive errors. Overall, refractive surgeries are safe, predictable and present high rates of satisfaction. Nevertheless, the induced epithelial, stromal and nerve damage alters corneal integrity and function, triggering a regenerative response. Complications that arise from corneal wound healing process might directly impact on visual outcomes of keratorefractive procedures. Most of these complications can be prevented or effectively treated with minimal consequences and minor impact on optical quality. Nevertheless, it is crucial to accurately and timely identify these corneal regeneration-related complications for successful counseling and management. Optometrists, as primary eye care providers, play an essential role in detecting anatomic and functional alterations in vision. It is therefore of great interest for optometrists to be familiar with the principal postoperative complications derived from alterations in regenerative process after corneal laser refractive surgeries. This review aims to provide a basis for optometrists to better understand, identify and manage the main wound healing-related complications after refractive surgery.
La cirugía refractiva corneal se lleva a cabo a menudo para corregir de manera permanente los errores refractivos. En general, la cirugía refractiva es segura y predecible, y presenta altos índices de satisfacción. Sin embargo, el daño inducido a nivel epitelial, estromal y nervioso altera la integridad y la función de la córnea, y desencadena una respuesta regenerativa. Las complicaciones que surgen del proceso de cicatrización de la herida corneal podrían tener un impacto directo sobre los resultados visuales de los procedimientos queratorefractivos. La mayoría de estas complicaciones pueden prevenirse, o tratarse de manera efectiva con mínimas consecuencias y un menor impacto sobre la calidad óptica. Sin embargo, es esencial identificar de manera precisa y oportuna dichas complicaciones relacionadas con la regeneración de la córnea, en aras de llegar a un asesoramiento y tratamiento satisfactorios. Los optometristas, al ser profesionales sanitarios de atención ocular primaria, juegan un papel esencial a la hora de detectar las alteraciones anatómicas y funcionales de la visión. Por tanto, es muy interesante que los optometristas estén familiarizados con las complicaciones postoperatorias principales derivadas de las alteraciones del proceso regenerativo tras cirugía refractiva corneal láser. Esta revisión tiene como objetivo proporcionar una base a los optometristas para la mejor comprensión, identificación y tratamiento de las principales complicaciones relacionadas con el proceso de cicatrización tras una cirugía refractiva.
Corneal refractive surgery is a rapidly evolving area of eye care. In general, refractive corneal surgery is successful and presents high levels of satisfaction,1–3 as a result of excellent uncorrected visual acuities and minimal residual defects.
Despite the success of laser refractive surgery to correct low order aberrations, the incidence of postoperative symptoms remains to be relatively high. The most common complaints are glare (27–55%), halos (30–55%) and reduced night vision (12–57%).1–3 These visual outcomes can reduce the quality of life of patients and are usually attributed to the induction of high order aberrations, particularly spherical aberration and coma, and large pupil diameters.4 However, this might be also the consequence of biological events in corneal wound healing process and due to pre- or intraoperative technical errors.5–9 Technical errors can be potentially avoided with an exhaustive selection of patients or with the development of new technologies. In fact, remarkable efforts are gone to refine and improve technology in laser refractive surgery. The evolution of technology and surgical techniques in the past decades has considerably reduced the incidence of visual disturbances, mainly by accelerating postoperative recovery. However, some drawbacks intrinsic to the procedure still persist unresolved. If the damage to corneal epithelium and stroma triggers an exaggerated wound healing response, it might culminate in undesirable consequences.6–8,10 In this context, an early detection, accurate diagnosis and treatment of complications are extremely relevant.
The optometrist, as primary eye care provider, plays a key role in the detection of potentially harmful complications of refractive surgery. Therefore, it demands the eye care professionals to be attuned to the principal postoperative complications derived from corneal refractive surgeries, and to recognize their symptoms and visual side effects to improve counseling and management of patients. This review provides a useful base for the diagnosis of corneal regeneration-related complications after corneal laser refractive surgery, and specifically deals with timings and treatments that are tailored to a particular complication in an attempt to contribute to a clearer understanding of wound healing complications for primary eye care professionals.
Corneal laser refractive surgeriesThe most commonly performed corneal refractive surgical procedures are Photorefractive Keratectomy (PRK) and Laser-Assisted in Situ Keratomileusis (LASIK). Briefly, PRK is a surface ablation procedure in which corneal epithelium is removed by mechanical debridement. With the evolution and refinement of PRK Advanced Surface Ablation (ASA) procedures emerged. In ASA procedures, corneal epithelium is removed by alcohol-assisted debridement (LASEK) or with an especial microkeratome, named epikeratome (epi-LASIK). An excimer laser in then used to ablate central corneal stroma in myopic corrections and peripheral corneal stroma in hyperopic corrections.7 In ASA procedures, the epithelial flap can be either discarded during the procedure (flap-off) or reapplied to the photoablated stromal bed (flap-on). The latter has been suggested that reduces postoperative pain and achieves faster visual recovery, but there is no consensus about the clinical superiority of this approach. LASIK is a lamellar procedure in which a corneal flap is created with a microkeratome or femtosecond laser and repositioned after applying excimer laser within the stroma.6 Small-Incision Lenticule Extraction (SMILE) is a recently introduced flap-less refractive surgery procedure. In this technique, the femtosecond laser creates an intrastromal corneal lenticule, which is extracted through a 2–5mm incision, usually in the upper peripheral cornea.8
Corneal refractive surgery mainly acts on the most anterior cornea, whereas mid and posterior stroma, Descement's membrane and corneal endothelium are presumably preserved.11 Corneal thickness and curvature are remodeled for inducing refractive changes, and because of that corneal anatomy is altered in different ways depending on the refractive procedure.11 So, the corneal response might considerably differ among surface ablation procedures, LASIK and SMILE.6–8
Corneal laser surgery-induced regenerative responseCorneal wound healing is a complex process that is regulated by the interaction between epithelial and stromal cells, nerve fibers and the tear film.12 Corneal regeneration response usually starts with epithelial injury. In corneal refractive surgery epithelial damage is caused by microkeratome blade, alcohol exposure, mechanical scraping, or femtosecond laser. Following this damage, epithelial cells release several molecules, such as interleukin-1 (IL-1), epidermal growth factor (EGF) or platelet-derived growth factor (PDGF) that contribute to the maintenance and restoration of the cornea.5,12–15 After the epithelial damage, stromal cells or keratocytes segregate cytokines to modulate the proliferation, migration and differentiation of epithelial cells and to repair the stroma.12
Epithelial and stromal responseThe breakdown of epithelial basement membrane is necessary to allow the interaction of molecules between epithelial and stromal cells. When epithelial-derived cytokines reach the stroma, they activate a cascade of events, including keratocyte apoptosis, proliferation and differentiation into myofibroblasts.16 The number of keratocytes that undergo apoptosis varies between refractive procedures and have been demonstrated by means of histological and in vivo studies.13 The initial keratocyte depletion is more pronounced in surface ablation procedures than in LASIK or SMILE, probably because in flap procedures the epithelium is preserved. Confocal microscopy has shown that in eyes that undergo PRK there is depletion of keratocytes under the treated region, and this density diminishes in a time period of 5 years with an approximate loss of 5% per year.17 Corneas treated with LASIK also show a continuous decrease in the density of keratocytes, although this seems to be less notorious than in surface ablation procedures.17 According to the results of Cañadas et al.,18 keratocyte density in the flap and stromal bed was decreased 15 months after LASIK and remained stable thereafter. Although decreased keratocyte density was observed, the differences were not significant when compared to control group, and were independent of the use of microkeratome or femtosecond laser for flap creation. Dong et al.,19 compared wound healing and inflammatory responses after SMILE and femtoLASIK techniques in rabbits, and lower apoptotic stromal cells were found in SMILE-treated corneas at 4 and 24h postoperatively and less proliferating cells at day 3 and week 1 after surgery with SMILE technique.19 Epithelial molecules also stimulate the production of collagenases and metalloproteinases from keratocytes to degrade damaged extracellular matrix and activate keratocytes to secrete proteoglycans and collagen fibrils to form the new extracellular matrix.
Some active keratocytes can differentiate into myofibroblasts. These cells have contractile ability and generate adhesion structures with the surrounding substrate.16 This is probably the first biological event for corneal haze formation.16 The fibrotic response is usually stronger after surface ablation procedures than in lamellar procedures, because subepithelial haze mainly occurs after epithelial manipulation or epithelial defects.20 The epithelial cell damage in LASIK is mainly restricted to the flap edge. In SMILE only a small peripheral incision of 2–5mm is created by femtosecond laser, so the epithelial damage is minimal. In an attempt to preserve the basement membrane and its attachment to the epithelium in surface ablation procedures, Pakillaris et al.,21 introduced epi-LASIK technique. Epi-LASIK microkeratome incorporates a suction ring and a blunt oscillating blade to rapidly separate the epithelium from the stroma.
Regeneration of subbasal nerve plexusThe disruption of corneal nerves is characteristic in each procedure. Along with corneal neural impairment, patients show altered corneal sensitivity and dry eye symptoms. In surface procedures, the photoablation disrupts intraepithelial terminals, subbasal plexus and anterior stromal nerves.17 In LASIK, the subbasal plexus on the borders of the flap and stromal nerves are axotomized, and remnants of subbasal plexus persist.13,22 On the other hand, long ciliary nerve and subbasal nerve denervation is less severe in SMILE.13,22 Regardless of the refractive procedure, there is corneal nerve disruption and this interrupts the corneo-lacrimal function. The receptors situated in corneal terminal nerves transmit the signal to segregate tears, and in their absence the lacrimal production decreases causing alterations in ocular surface and dry eye symptoms.17,23,24 Corneal nerve disruption, therefore, produces ocular dryness and altered corneal sensitivity. Besides, axotomy of corneal nerves triggers a response for axon fragmentation, the removal of the debris, and the release of inflammatory mediators, such as histamine and serotonin.25 All of these events lead to a regenerative state, in which a transient nervous plexus arises until complete axonal restoration is concluded.26 The process culminates with the pruning of nerve sprouts, but if the process of regeneration is interrupted, nerve sprouts can evoke sensations of burning, foreign body sensations or sand-like feeling.26,27
Ocular sensitivity and dry eye syndromeDry eye is considered one of the most common complications after corneal laser refractive surgery, and sensory denervation seems to be the major causal factor.26 After PRK, central corneal nerve recovery may last up to two year, as demonstrated by a 5-year longitudinal study.17 The authors observed that subbasal nerve density decreased in 59% in the first year after PRK and was completely recovered at two years. According to the same authors,17 after LASIK nerve recovery took longer, and a 34% decrease was found in the density of subbasal plexus at 3 years and preoperative values were achieved at 5 years. In SMILE technique, a different pattern of neural damage is induced, and the reduction of nerve fiber density is likely less severe than in other corneal refractive surgeries.8,22,28,29 Denoyer et al.,28 found that the number of long fibers and its ramifications were greater in the SMILE group than in LASIK group at 1 month and 6 months after surgery, which is consistent with the results of Mohamed-Noriega y et al.,22 On the other hand, Liu et al.,29 found that subbasal nerve density after SMILE gradually recovered until preoperative values were found at 3 months.
Nerve fiber axotomization alters corneal sensitivity and tear film stability that slowly recovers in the following postoperative months to years.7,15,22,26 The recovery of corneal sensitivity in PRK usually starts at 4–6 postoperative weeks, although a complete recovery of sensitivity – near to preoperative values – may take as long as a year.17 Regarding corneal sensitivity in LASIK, it recovers in the first 6 to 12 months.30 Nonetheless, Gallar et al.,31 determined by means of gas esthesiometry that preoperative values of corneal sensitivity after LASIK were obtained at 2 years. LASIK-induced neurotrophic epitheliopathy is described as a severe dry eye, believed to result from the section of the long ciliary nerves accompanied by a poor nerve regeneration which hinders sensory corneal feedback.23 Aberrant or irregular nerve regeneration might also culminate in neuropathy, leading to persistent and intense corneal pain sensations.26,27 Faster corneal reinnervation after SMILE probably explains why corneal sensitivity is recovered sooner in SMILE than in LASIK.9
During corneal nerve recovery period, patients show altered corneal sensations and clinical signs. According to the study of Hovanesian et al.,24 from 251 patients that had undergone PRK surgery in the previous 6 months, a 43% reported dry eye symptoms that were more evident after awakening. As reported by Lee et al.,32 6 months after PRK Schimer test values were diminished 14.57±6.39%, tear break-up time (BUT) values 12.54±8.28%, and tear osmolarity was increased in 14.95±6.46%. At 12 months after PRK, 5% of patients present chronic dry eye disease.33 Similarly, Ganesh and Gupta34 observed that Schimer test and BUT values were decreased 3 months after LASIK. LASIK-induced dry eye signs and symptoms disappear with the recovery of corneal sensation, usually between 6 months and one year.30 This period is much shorter than corneal nerve fiber regeneration.17 Same authors also observed that 3 months after SMILE Schimer test and BUT values were significantly better in SMILE-treated eyes that in femtoLASIK-assisted patients.34 Throughout the recovery period, approximately 10% of patients refer fluctuating vision and have reduced best distance corrected visual acuity.23 Reported visual symptoms include visual fatigue or glare sensitivity.35
Post-refractive surgery dry eye management is similar to dry eye management of other etiologies. Artificial tears are usually the first line of treatment, but they can be combined with autologous serum drops, cyclosporine A, punctal plugs or protective glasses in more severe cases.23
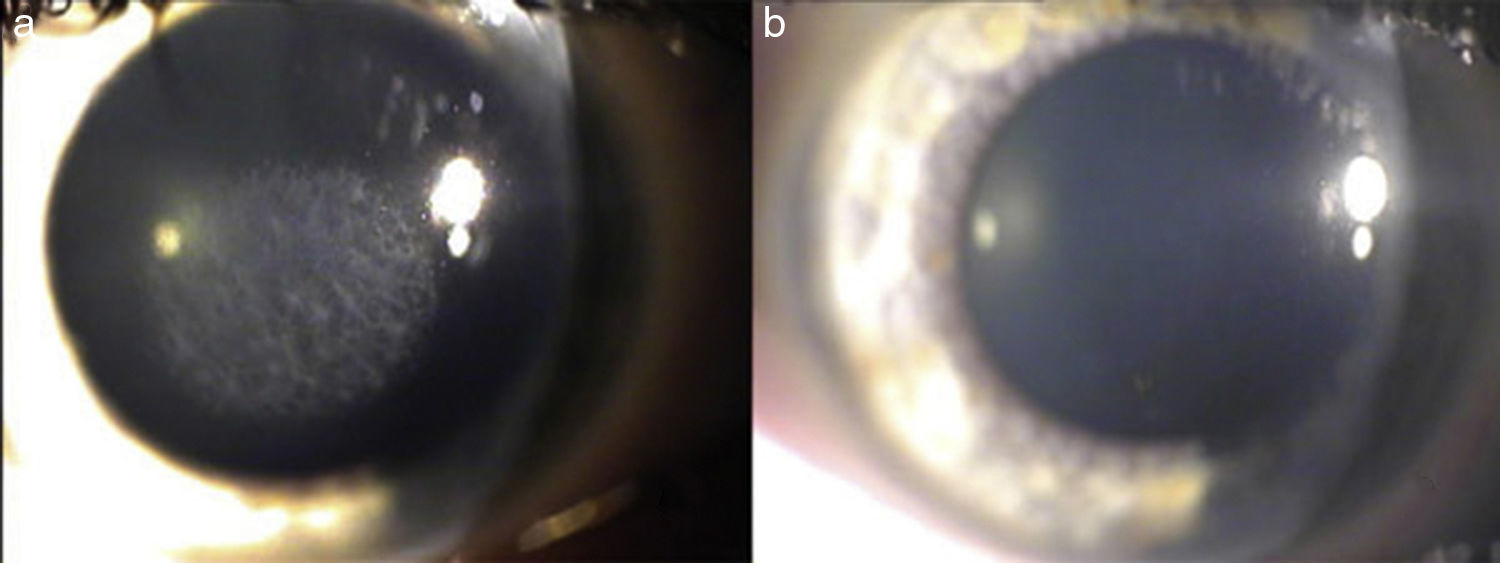
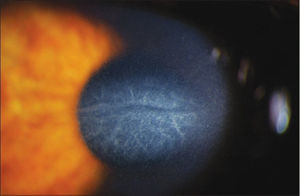
Regeneration-related complications: visual outcomes and managementCorneal hazePostoperative corneal haze is an anterior stromal opacity that appears usually in the first weeks or months after PRK (Fig. 1). Although rarely, late-onset haze has been described too. Surgery-induced damage to the basement membrane results in the abnormal deposition of collagen fibers in subepithelial area that contributes to decreased corneal transparency.16 This type of corneal haze is located typically at the subepithelial level, is secondary to myofibroblast formation from activated keratocytes, and should be differentiated from corneal opacification at deeper locations, which may be associated to a variety of inflammatory conditions or wound healing responses unrelated to surface ablation procedures on a virgin cornea. Myofibroblasts segregate extracellular matrix, contract wounds and form adhesion structures with the surrounding tissue.16 Some active keratocytes seem to be transformed in myofibroblasts in the presence of tear film-derived tumoral growth factor-β1 (TGF-β1).16 For that to happen, an altered or absent basement membrane is necessary, as it happens in PRK. Epi-LASIK was introduced to preserve the basement membrane and its attachment to the epithelium in surface ablation procedures. However, a recent meta-analysis by Wen et al.,36 has failed to find any differences between epi-LASIK and PRK in terms of safety, predictability, efficacy and visual quality. As keratocytes are minimally altered after SMILE, it is presumed that the incidence of corneal haze is lower than in other corneal refractive surgeries. In fact, Ivarsen et al.,37 demonstrated that only 8% of 1800 SMILE-treated eyes developed corneal haze.
Subepithelial haze occurs in the majority of patients 1 month after PRK, reaching the greatest intensity between 3 and 6 months postoperatively, and gradually decreases thereafter.20 Some authors claim that it can remain as long as 12–24 months.38 After that period, subepithelial corneal haze considerably decreases spontaneously.20 Subepithelial haze is more common when there is an abrupt curvature change between the ablated area and surrounding tissue, such as in myopic corrections above 6 diopters, hyperopic corrections higher or equal to 4 diopters and in high astigmatic corrections.16,39
The first line of treatment for corneal haze is prevention.7 Pharmacological agents that modulate wound healing response, like mytomicin-C (MMC), are topically administered intraoperatively to avoid or, at least, minimize myofibroblast activation.40 Although prophylactic antibiotics are prescribed to prevent corneal infections after refractive surgery, it has been demonstrated that fourth generation fluoroquinolones also enhance corneal healing rate.41 A more recent study has shown that Plasma-Rich in Growth Factors (PRGF) protects and reverses the myofibroblast phenotype while promoting cell proliferation and migration.42 Antiinflamatory drugs are regularly prescribed after refractive surgery to reduce pain and inflammation. Certain non-steroidal drugs are known to delay corneal reepithelization, which might increase the risk of haze formation. Nevertheless, it seems that steroidal antiinflamatory drugs rather reduce the risk of short-term corneal haze formation.43 Haze development is also related to the duration of epithelial defects, male sex, and excessive exposure to UV-B.7 UV-B radiation exacerbates and prolongs stromal healing response. Therefore, the use of UV-protective eyewear should be encouraged during the first 6 postoperative months. All these preventive measures are usually sufficient to minimize the incidence of corneal haze. However, in rare cases when visually significant corneal haze develops, it can be successfully treated with phototherapeuthic keratectomy (PTK) with MMC application.
Corneal haze appears in the first weeks or months after PRK, and low contrast visual acuity might be slightly reduced during this period. It can also provoke nocturnal symptoms that usually improve with time. If corneal haze persists over time, it may produce irregular astigmatism and decreases corrected visual acuity.20 The incidence of clinically significant corneal haze was as high as 2–4% in past excimer laser procedures,39 but with the advent of new excimer lasers and appropriate preventive treatments such as intraoperative MMC application, the incidence of corneal haze has considerably dropped.
Epithelial hyperplasia and regression of refractive errorMild regression of refractive error has been described after PRK and LASIK procedures.20,44 In high myopic patients (more than 6 diopters), regression occurs in 78% of patients one year after PRK,20 and virtually in all hyperopic eyes.44,45 This refractive regression seems to be a direct consequence of epithelial hyperplasia. The cornea tends to regularize corneal imperfections to even corneal surface, which is extremely important for maintaining the optical quality.44 As epithelial hyperplasia is relatively common after refractive procedures, this expected corneal response is already considered in standard nomograms to perform the surgery. The increase in epithelial thickness due to abnormal keratocyte density and subepithelial deposits are significantly associated with myopic regression.44 The increase in epithelial thickness is more persistent and prolonged after PRK than in LASIK46 – it stabilizes 1 week after LASIK and 1 year after PRK.46 Histological studies in animals and humans have observed that the increase in epithelial thickness is due to an elongation of basal epithelial cells and to an increase in superficial cells.47 Although SMILE is a minimally invasive procedure, Ivarsen et al.,48 reported 5 cases of epithelial hyperplasia and irregular Bowman's layer after SMILE that required retreatment with PRK to enhance optical quality. Vestergaad et al.,49 also found epithelial hyperplasia after SMILE for moderate to high myopic corrections.
Excessive epithelial hyperplasia can reduce the refractive outcomes of corneal refractive surgery.20,44 When the actual corneal hyperplasia is greater than the forecasted response, residual errors can be corrected by means of ophthalmic lenses, contact lenses or refractive surgery if normal post-surgery topography and stable refractive error are found for at least 3 months.
Interface complicationsLASIK creates a new anatomic space between the flap and the underlying stroma that is referred to as the interface.10 Therefore, interface complications are almost exclusive of this technique.10 In this space an accumulation of cellular debris or inflammatory cells can occur, or even the buildup of liquids that sometimes are presented with overlapping clinical signs and symptoms. Epithelial ingrowth, diffuse lamellar keratitis (DLK), interface fluid syndrome, and central toxic keratopathy (CTK) are the main interface complications that will be discussed in this section.50–53
Epithelial ingrowth can only occur after lamellar procedures. In primary LASIK, epithelial ingrowth is relatively rare, but the incidence greatly increases when lift-flap LASIK retreatment is performed two years or longer after the primary LASIK, or with the use of contact lenses after retreatment.10 It is most commonly detected at 1-month as white, milky deposits at the level of the peripheral interface.50 It is a normal response to LASIK surgery,13 and associated visual symptoms are minimal.50 These cases are considered clinically insignificant and are managed with observation.54 However, epithelial ingrowth can become an aggressive complication if extended to visual axis, as it can decrease best corrected visual acuity, provoke glare if deposited in pupil edge, or induce irregular astigmatism if focal flap elevation takes place.10 Treatment is required when epithelial ingrowth obstructs the visual axis. Usually lifting and scraping stroma anterior and posterior to the interface is sufficient.10
Diffuse lamellar keratitis (DLK) is an early postoperative complication due to the infiltration of inflammatory cells between the flap and stromal bed.51 Several factors can induce white blood cell infiltration. In sporadic DLK, the main causes are usually intraoperative epithelial cell damage, atopic disease or Meibomian gland dysfunction, whereas in epidemic DLK, it is mostly related to biofilm reservoirs in surgical instruments, being gram-negative endotoxins the triggering agents.51,55 Late onset DLK has also been described in the literature and can occur anytime after LASIK following epithelial injury.10
Visual impairment depends on the severity of the condition. In most cases, DLK tends to resolve with steroid treatment.55 After resolution, uncorrected and corrected visual acuities are satisfactory.51,56 In stages 1 and 2, central corneal is spared, whereas in stages 3 and 4 the infiltration is situated in visual axis decreasing visual acuity.51 Stage 3 decreases visual acuity in 2 or 3 lines. Stage 4 is extremely rare, and it is characterized by dense scarring, and can permanently decrease visual acuity.10 DLK induces hyperopic shift and might be accompanied by regular or irregular astigmatism, depending if corneal melt takes place.51 It seems that DLK alters contrast sensitive more than visual acuity. Han et al.,57 observed similar visual acuities in DLK and non-DLK eyes, but contrast sensitivity was significantly reduced in DLK group. Even though interface complications are almost exclusive of LASIK, DLK has also been described after SMILE, but the incidence is much lower.58 According to Zhao et al.,58 1112 patients underwent SMILE surgery and only 18 patients developed DLK. Unlike in LASIK, lenticule thickness is the unique factor contributing to the onset of DLK after SMILE.58 The most common signs of DLK are ocular hyperemia and corneal infiltrates that are accompanied by symptoms of tearing and pain.58
Fluid accumulation of variable degree can occur in the interface. Galvis et al., proposed to encompass those conditions under the name of post-LASIK edema-induced keratopathy (PLEK).59 Interface edema is rare beyond 1 week post-LASIK and can occur in patients with Fuch's corneal dystrophy, in patients with high intraocular pressures secondary to steroid treatments post-LASIK (we refer to this condition as pressure-induced stromal keratitis) or in patients in treatment with drugs that slow endothelial pumping function (e.g. carbonic anhydrase inhibitors).60,61 Interface fluid buildup can range from a clinically imperceptible diffuse haziness52 to a visible severe fluid accumulation that separates the flap from the stromal bed.62 In early stages of PLEK, the signs and symptoms are very similar to DLK, and a diligent diagnosis is vital. If correctly managed, the best corrected visual acuity is recovered.52,62 However, it has been documented that late diagnosis can conclude in glaucomatous loss of visual field and decreased visual acuity.63
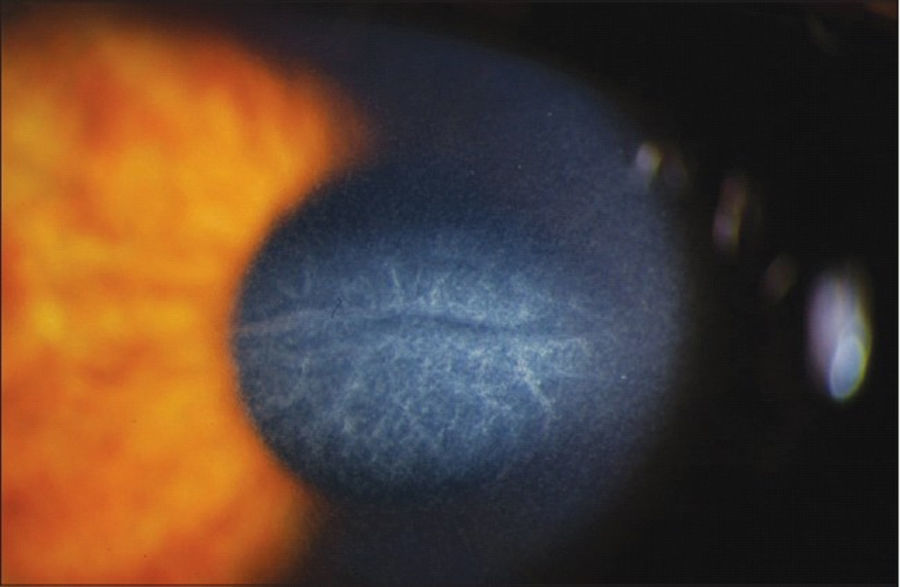
Central toxic keratopathy (CTK) is presented as a central corneal opacity accompanied by stromal tissue loss after LASIK53 (Fig. 2). Its etiology is still unknown, although it has been proposed that uncontrolled enzymatic degradation during wound healing process might be the pathological substrate. CTK is a rare non-inflammatory condition that presents acutely and is painless.53 Anterior segment tomography has shown corneal surface flattening with consequent hyperopic shift.64 It is a self-limited condition that usually resolves within 18 months and most patients recover visual function, even though a faint corneal haze and hyperopic shift may persist in over time.53,64
Central Toxic Keratopathy with loss of central corneal transparency. Image reproduced under Creative Commons Attribution License. Hazin R et al., 2010. Middle East Afr J Ophthalmol. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2880375/.
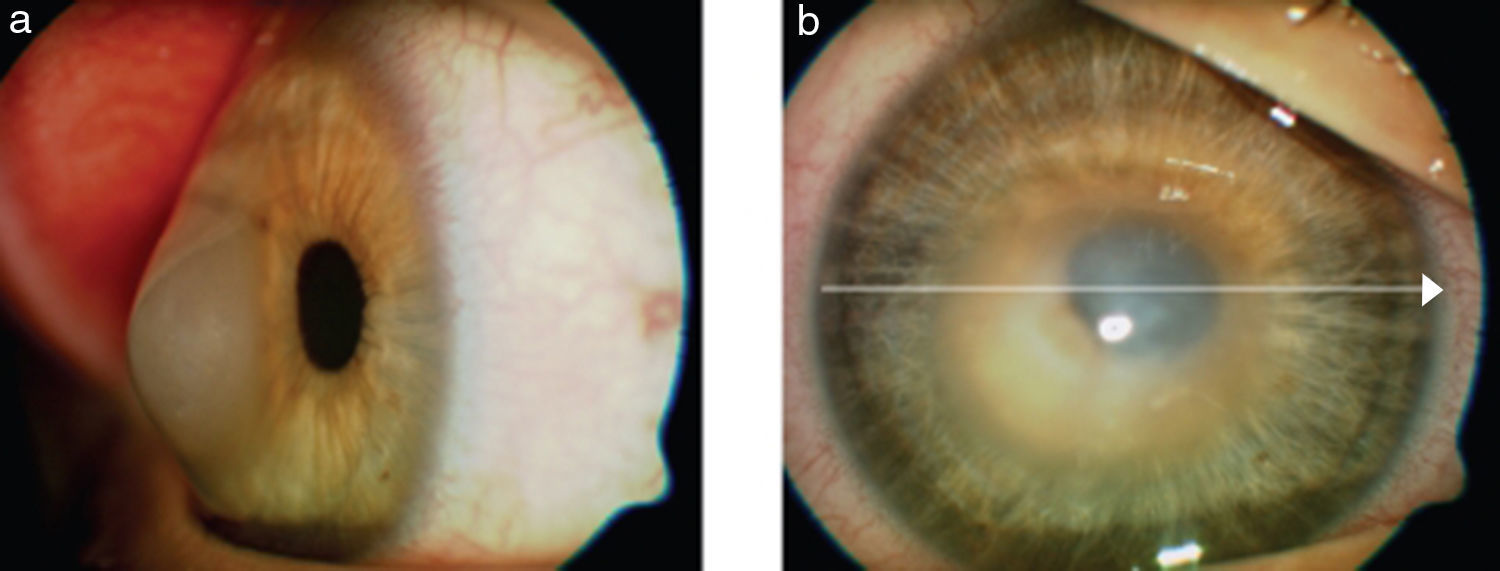
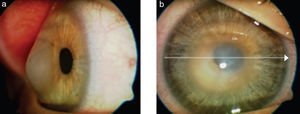
Corneal ectasia is a rare complication that can occur after refractive surgery. It is a progressive steepening and thinning of the stromal tissue accompanied by vision impairment65,66 (Fig. 3). Its onset varies from 1 month to several years after refractive surgery.67 The real incidence is unknown, but it is estimated to be between 0.03% and 0.6% after PRK and LASIK.65,66 El-Naggar.,68 casuistically informed of one case of keratoectasia after SMILE, but probably because a frustrated form of keratoconus was previously present.
In this image we can appreciate post-LASIK keratectasia with cornea budged forwards and edematous central cornea. Image reproduced under the CC-NY License. Meyer H et al., 2009. J Ophthalmol. http://www.ncbi.nlm.nih.gov/pubmed/20339447.
Although the etiology of the ectatic process is still to be elucidated, some authors have suggested that a failure in corneal regeneration might be the cause.69,70 According to this hypothesis, active stromal remodeling would not conclude and therefore ongoing keratocyte activation would be constantly releasing intracellular components, like degradative enzymes that could potentially damage corneal stroma, decreasing its tensile strength.69,70 Further studies need to be carried out to confirm this hypothesis. So far, several risk factors have been identified, such as younger age, abnormal preoperative topographies, thin corneas, high myopias, and low stromal bed thicknesses,71 but the percentage of altered tissue is the most predictive factor for developing corneal ectasia.72,73 Another relevant risk factor associated to the onset of post-refractive surgery ectasia is eye rubbing, an understated factor that the optometrists can help to diminish effectively by delivering appropriate advice and counseling.71
By definition, corneal ectasia reduces vision and is accompanied by an array of symptoms, including glare, ghost images or double vision. Reduced visual performance is secondary to refractive changes and increased high-order aberrations, particularly vertical coma.74 Ectatic corneas usually present a myopic shift, because of central corneal protrusion, with increased astigmatism that might become irregular as the corneal bulge increases.72 This leads to reduced best-spectacle corrected visual acuity that highly correlates with the degree of ablated tissue.72 To date, cross-linking is the unique treatment capable of stabilizing the progression of ectatic diseases, but might not be successful as the sole treatment for improving visual acuities and refractive status.75 Therefore, cross-linking is combined with rigid or scleral contact lenses and/or intrastromal rings for regularizing anterior surface of the cornea.
ConclusionsMany efforts are underway to prevent complications and improve the outcomes of refractive surgery. Recent advances in ophthalmic surgery and the evolution of technologies have decreased the probability of postoperative complications associated to corneal laser refractive surgery. An increased awareness of the contraindications has also resulted in fewer postoperative complications. Nevertheless, morphological and functional alterations on corneal surface persist. The triggering of wound healing process promotes biological events that are mostly unpredictable, and controlling them is still an endowing tasks. Postoperative complications such as under and overcorrections or corneal opacities are largely related to corneal regeneration. History and onset of symptoms accompanied by a careful clinical examination allow accurate diagnosis and treatment on presentation, and will determine the ultimate visual outcomes. It is therefore imperative to identify each of these conditions. Optometrists, as primary eye care professionals, must be well educated on these potential complications and challenges to provide refractive surgery patients appropriate management and referral.
Conflicts of interestThe authors have not proprietary or commercial interest in the medical devices that are involved in this manuscript.