To assess the prevalence of esophoria at near in concussed patients in a neuro-optometric private practice setting.
MethodsA retrospective analysis was performed involving a chart review in a neuro-optometric, private practice setting of consecutive patients with a medical diagnosis of concussion from January 1st 2016 to December 31st 2016. A total of 71 patients were included in the analysis. All received a comprehensive vision examination, with a near vision emphasis. The near horizontal phoria was assessed with the cover test and the von Graefe test.
ResultsApproximately 30% of the patients with a medically based diagnosis of concussion exhibited esophoria at near, with good agreement (95%) between the two tests. Mean esophoria was 5.2 (SD=2.8) prism diopters (pd), with a range from 2pd to 14pd of esophoria. Convergence excess was diagnosed in 23%.
DiscussionNear esophoria was found in nearly one-third of this practice-based sample of concussed patients. Thus, it was more common than typically believed to be the case. Two proposed oculomotor-based mechanisms related to these symptomatic esophoric patients included phoria decompensation and excessive accommodative vergence.
Evaluar la prevalencia de la endoforia de cerca en pacientes con conmoción, en un centro privado de neuro-optometría.
MétodosSe realizó un análisis retrospectivo, en un centro privado de neuro-optometría, con revisión de las historias de pacientes consecutivos con diagnóstico médico de conmoción desde el 1 de enero al 31 de diciembre de 2016. Se incluyó en el análisis a un total de 71 pacientes. A todos ellos se les realizó un examen ocular amplio, enfatizando la visión de cerca. La foria horizontal de cerca se valoró mediante el cover test y la prueba de von Graefe.
ResultadosAproximadamente el 30% de los pacientes con diagnóstico médico de conmoción mostraron endoforia de cerca, con buena concordancia (95%) entre las dos pruebas. La endoforia media fue de 5,2 (SD=2,8) dioptrías prismáticas (pd),con un rango de 2pd a 14pd de endoforia. El exceso de convergencia se diagnóstico en el 23%.
DiscusiónSe encontró endoforia de cerca en cerca del 25% de la muestra de pacientes con conmoción. Por tanto, esta condición clínica resultó ser más común de lo que se creía. Dos mecanismos propuestos basados en el sistema oculomotor con relación a estos pacientes endofóricos sintomáticos incluyeron descompensación de foria y exceso de vergencia acomodativa.
The visual sequelae of having received a concussion are manifold. For example, many patients report problems with eye movements, reading, visual attention, accommodation, visual motion sensitivity, and other visually related aspects.1 The presence of one or more of these vision problems is likely to have adverse consequences on one's activities of daily living (ADL), i.e., vocational and avocational activities and tasks.1
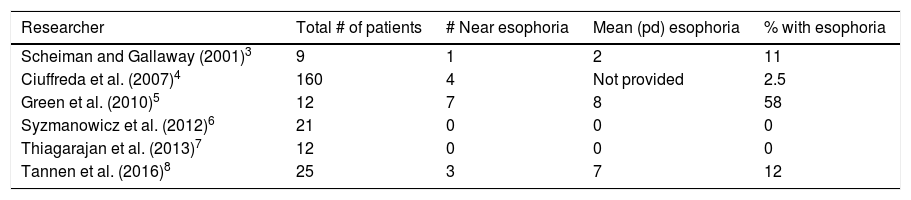
Conventional “clinical wisdom” suggests that many patients with concussion will typically manifest a large exophoria at near, that is one greater than 6 prism diopters (pd) or so, likely in conjunction with convergence insufficiency (CI).2 However, a careful assessment of several relatively recent studies points to a somewhat different picture, with a much wider range of possibilities. Table 1 presents the key, relevant findings in several current, well-done studies typically performed in academic, clinical environments.3–8 The range was very wide, namely 0–58%, with a mean of 14%. Such a large range makes diagnostic, predictive modeling difficult, especially for the clinician.
Prevalence of esophoria at near in concussion/mTBI patients: recent studies.
| Researcher | Total # of patients | # Near esophoria | Mean (pd) esophoria | % with esophoria |
|---|---|---|---|---|
| Scheiman and Gallaway (2001)3 | 9 | 1 | 2 | 11 |
| Ciuffreda et al. (2007)4 | 160 | 4 | Not provided | 2.5 |
| Green et al. (2010)5 | 12 | 7 | 8 | 58 |
| Syzmanowicz et al. (2012)6 | 21 | 0 | 0 | 0 |
| Thiagarajan et al. (2013)7 | 12 | 0 | 0 | 0 |
| Tannen et al. (2016)8 | 25 | 3 | 7 | 12 |
Thus, the purpose of the present retrospective study was to determine the frequency of occurrence of esophoria at near in a neuro-optometric, private practice that serves as a regional referral center for concussed patients with ongoing visual symptoms. These patients had medical documentation of the concussion, and all received a subsequent comprehensive vision examination by the first two authors.
MethodsThe clinical records of consecutive patients from January 1st to December 31st 2016 with a medical diagnosis of concussion were reviewed from the first author's private practice. All patients were referred from physicians who specialized in concussion management and were visually symptomatic (e.g. blur at near, visually related headaches). Patients with strabismus, amblyopia, ocular disease, developmental disabilities, neurologic disease, or psychiatric disorders which did not exist prior to the initial concussion were excluded. The combined ocular health/neuro-ophthalmic examination, which included tonometry, screening visual fields, external examination, pupil evaluation, slit lamp examination, and dilated fundus exam, was unremarkable in all of the patients.
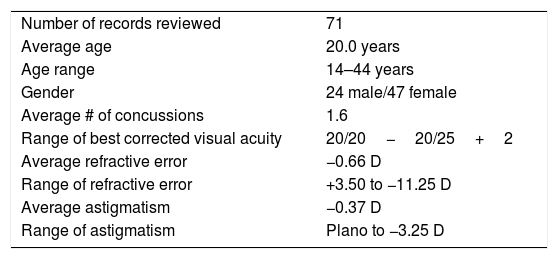
Seventy-one patients met the above criteria. Ages ranged from 14 to 44 years, with a mean of 20.0 years. There were 24 males and 47 females. Their most recent concussion was diagnosed from 2 to 11 months prior to the vision examination/consultation, with the average time being 4.3 months. Table 2 is a summary of the basic demographic, refractive error, and visual acuity data for these patients.
Demographic data for patients included in record review.
| Number of records reviewed | 71 |
| Average age | 20.0 years |
| Age range | 14–44 years |
| Gender | 24 male/47 female |
| Average # of concussions | 1.6 |
| Range of best corrected visual acuity | 20/20−20/25+2 |
| Average refractive error | −0.66 D |
| Range of refractive error | +3.50 to −11.25 D |
| Average astigmatism | −0.37 D |
| Range of astigmatism | Plano to −3.25 D |
Table 3 presents a summary of the near tests performed on these patients. All testing was conducted with the patient's habitual distance spectacle prescription in place, unless the new distance refraction indicated a change, or a near prescription was deemed appropriate. All tests were performed using standard clinical guidelines/protocols.9 Details of the testing are provided elsewhere.8–10
Near vision tests included in the analysis.
| Clinical test |
|---|
| Near lateral phoria (pd) |
| Near cover test (pd) |
| NPC break (cm) |
| NPC recovery (cm) |
| Near base-in blur/break/recovery (pd) |
| Near base-out blur/break/recovery (pd) |
| Vergence facility (cpm) |
Symbols: pd=prism diopters, D=diopters, cm=centimeters, cpm=cycles per minute.
Additional testing aspects included the following. With respect to the near horizontal cover test, fusion was disrupted for at least 10s to allow the full phoria to become manifest.6 For the diagnosis of convergence excess and convergence insufficiency, the criteria of Master et al.2 were followed. These criteria are presented in Table 4.
Criteria for convergence insufficiency and convergence excess.a
| Diagnosis | Criteria |
|---|---|
| Convergence insufficiency | Near point of convergence of 6cm break and - Reduced positive fusional convergence at near (<20 pd or fails Sheard's criterion) or - Vergence facility (distance or near) ≤9cpm with more difficulty with base-outa |
| Convergence excess | ≥3 pd esophoria at near and - Reduced negative fusional convergence at near (<8 pd or fails Sheard's criterion) or - Vergence facility at near ≤9cpm with difficulty with base-ina |
Table 5a presents the results for the cover test at near. Twenty-eight percent of the patients exhibited esophoria at near. The mean was 5.2 (2.8) prism diopters (pd), with a range from 1 to 14pd. In contrast, 72% of the patients exhibited exophoria at near. The mean was 8.2 (3.4) pd, with a range from 3 to 16pd.
Table 5b presents the results for the von Graefe phoria test at near. Thirty-one percent of the patients exhibited esophoria at near. The mean was 5.2 (3.4) pd, with a range from 2 to 12pd. In contrast, 69% of the patients exhibited exophoria at near. The mean was 8.0 (4.4) pd, with a range from 1 to 16pd. Of the twenty patients who showed esophoria on the near cover test, 19 (95%) also exhibited esophoria on the von Graefe phoria test at near; the remaining patient was orthophoric.
Table 6 presents the results for convergence excess (CE) versus convergence insufficiency (CI). Twenty-three percent manifested CE, whereas 77% manifested CI.
Convergence excess vs. convergence insufficiency (per diagnostic criteria in Table 4).
| N | % | |
|---|---|---|
| Convergence excess | 7 | 23 |
| Convergence insufficiency | 24 | 77 |
The findings of the present study reveal the high prevalence (∼30%) of esophoria at near, and being of considerable magnitude, in this neuro-optometric, office-based sample of individuals with concussion. The value of 30% is significantly greater than the mean value (∼14%) averaged across the earlier cited investigations (Table 1), but lies in their midrange (0–58% range). Furthermore, the prevalence of esophoria at near in four of the six studies cited (i.e., in those with other than zero values) is considerably greater (∼20%) than found in a large (n=4880) non-concussed, asymptomatic, primarily adult (∼90%) clinic population (7.6% esophoria).11
Why is there such a high prevalence of near esophoria in the present sample of concussed patients? There are at least three possibilities. First, by the process of self-selection, patients with a newly acquired esophoria at near may comprise those who do not resolve by natural recovery over time, remain visually symptomatic, and subsequently seek a comprehensive optometric vision examination, with likely visual intervention (e.g., oculomotor-based vision rehabilitation)12 to resume their vocational and avocational goals. Thus, they may eventually be examined at a specialty neuro-optometric practice. Second, and related to the first, those with prior asymptomatic, habitual near esophoria may manifest the phenomenon of phoria-based vergence “decompensation”13 as a result of their head injury. That is, their vergence adaptation mechanism14 has become impaired by the concussion(s), and thus their fusional motor-based compensatory ability is reduced. They now become visually symptomatic at near (e.g., intermittent diplopia, asthenopia). Third, it is likely that many of these individuals had not initially received a complete near vision evaluation, including the near phoria. If they were then referred to receive some “basic” vision therapy from either a physical or occupational therapist, the patient might have been treated for presumed exophoria and convergence insufficiency, as more commonly reported in the concussed population.2 Such therapy would then in fact be counter-productive, and furthermore likely to exacerbate the original visual symptoms at near.
The wide variation across studies is interesting. For example, in the four studies conducted in the Ciuffreda laboratory,4–7 the measurement techniques were similar and carefully performed. The test subjects (while derived at different times over a seven year period) were from the same academic clinical setting, yet the range varied from zero to fifty-eight percent having near esophoria. This variation might result from convenience sampling. It warrants further investigation over a range of large, different sample populations, such as hospital settings, the medical concussion specialist, and other optometric practices with an emphasis on the concussed patient, as different clinical settings may have unique distributions of near phoria as per their diverse referral sources. This information would help the predictive powers and diagnostic prowess of the clinical practitioner.
There may also be other reasons for these differences in studies. First, subjects in the present investigation were all in the late subacute-early chronic phase of mTBI,15 whereas those in the Ciuffreda studies4–7 were well into the chronic phase. Second, while subjects in all of the studies were visually symptomatic, type and/or severity of the visual symptoms were not controlled. The individual with the symptom of photosensitivity may differ markedly from one with intermittent blur at near. And, third, since all subjects in the present study were referred by a concussion specialist, these may represent the more symptomatically severe cases. Again, further studies in the area are warranted to unravel this complex situation.
What might be the related oculomotor mechanisms involved? There at least two possibilities. First, as mentioned earlier, phoria-based vergence decompensation may have occurred following the concussion, thus giving rise to near visual symptomatology.13 Second, as many of these concussed patients typically concurrently manifest an accommodative insufficiency (∼40%) following brain insult,4 the younger pre-presbyopic patient, as found in the present study, may attempt to exert volitional control of the accommodative system, hence in turn over-driving the accommodative vergence system due to their mutual interaction, and therefore resulting in esophoria at near and related asthenopia.
For clinicians who wish for a more systematic approach to the case history, evaluation, management, and conceptual underpinnings for the clinical care of concussion/mTBI patients, especially to “unveil” a concussion, we have included the appropriate references.10,16–18 Some critical case history questions toward this end include: what were the initial visual symptoms and their severity; how long did they last; and are there any remaining visual symptoms and related problems at today's vision examination, and if so, what are they, and how severe are they?
In conclusion, the findings of the present investigation reveal that the presence of esophoria at near in the concussed individual may be more common than typically believed to be the case, especially based on the earlier studies (Table 1). Thus, the near phoria must be carefully and fully evaluated by the clinician, as its presence has important ramifications diagnostically, prognostically, and therapeutically in the concussed individual.
Conflicts of interestThe authors have no conflicts of interest to declare.