This pilot study sought to determine the efficacy of using the Developmental Eye Movement (DEM) test in the adult, acquired brain injury (ABI) population to quantify clinically the effects of controlled, laboratory-performed, oculomotor-based vision therapy/vision rehabilitation.
MethodsNine adult subjects with mild traumatic brain injury (mTBI) and five with stroke were assessed before and after an eight-week, computer-based, versional oculomotor (fixation, saccades, pursuit, and simulated reading) training program (9.6h total). The protocol incorporated a cross-over, interventional design with and without the addition of auditory feedback regarding two-dimensional eye position. The clinical outcome measure was the Developmental Eye Movement (DEM) test score (ratio, errors) taken before, midway, and immediately following training.
ResultsFor the DEM ratio parameter, improvements were found in 80–89% of the subjects. For the DEM error parameter, improvements were found in 100% of the subjects. Incorporation of the auditory feedback component revealed a trend toward enhanced performance. The findings were similar for both DEM parameters, as well as for incorporation of the auditory feedback, in both diagnostic groups.
DiscussionThe results of the present study demonstrated considerable improvements in the DEM test scores following the oculomotor-based training, thus reflecting more time-optimal and accurate saccadic tracking after the training. The DEM test should be considered as another clinical test of global saccadic tracking performance in the ABI population.
Este estudio piloto trató de determinar la eficacia del uso de la prueba DEM (Developmental Eye Movement) en la población adulta con daño cerebral adquirido (DCA) para cuantificar clínicamente los efectos de la rehabilitación/terapia visual controlada, realizada en laboratorio, y de carácter oculomotor.
MétodosSe valoraron nueve sujetos adultos con daño cerebral traumático leve (mTBI) y cinco con ictus, con anterioridad y posterioridad a un programa de entrenamiento de ocho semanas, informático oculomotor y versional (movimientos de fijación, sacádicos, de persecución y lectura simulada, de 9,6 horas en total). El protocolo incorporó un diseño cruzado e intervencional, con y sin adición de retroalimentación auditiva en relación a la posición bi-dimensional de los ojos. La medida del resultado clínico se llevó a cabo mediante la puntuación de la prueba DEM (Developmental Eye Movement) (ratio, errores) realizada con anterioridad, en el punto medio, e inmediatamente tras el entrenamiento.
ResultadosPara el parámetro de ratio DEM, se encontraron mejoras en el 80-89% de los sujetos. Para el parámetro de error DEM, se encontraron mejoras en el 100% de los sujetos. La incorporación del componente de retroalimentación auditiva reveló una tendencia hacia la mejora del rendimiento. Los hallazgos fueron similares para ambos parámetros DEM, así como para la incorporación de la retroalimentación auditiva, en ambos grupos diagnósticos.
DiscusiónLos resultados del presente estudio demostraron mejoras considerables de las puntuaciones de la prueba DEM tras la terapia oculomotora, lo que refleja una mayor optimización y precisión del seguimiento sacádico en el tiempo tras la terapia. Deberá considerarse la prueba DEM como una prueba clínica para valorar el rendimiento del seguimiento sacádico global en la población de DCA.
The topic of “concussion” has been prominent over the past decade in both the medical literature and popular press, in part due to publicity in the sports arena, especially regarding football.1,2 It is estimated that there are 300,000 sports-related concussions annually in the United States.3 Moreover, there is evidence that after having incurred even a single concussion, the visual system can be adversely affected in 15% of these individuals.4 Further, a concussion can lead to visual system signs and symptoms of both a short- and long-term nature.5 Lastly, having a first concussion increases the probability of subsequent ones,6 perhaps due to reduced attentional capacity and impaired vigilance, as well as possible concurrent dizziness and vestibular problems.
The diagnosis of a concussion is critical in contact sports, such as football and hockey, where such decisions frequently are made rapidly on the sidelines based on simple verbal query and/or gross visual observation of the player. If not diagnosed properly, it could lead to a subsequent concussion in the same game, with potentially longer-term consequences, as mentioned earlier. Several ideas have been put forth regarding making a tentative sideline concussion diagnosis, including the use of objective pupillometry,7,8 cognitive testing,9–11 near point of convergence,12,13 distance vergence facility,14 and global saccadic eye movement assessment,10,15,16 with the last typically employing the King-Devick (K-D) test.11,17 The K-D test provides a simple, rapid, and quantitative manner for such assessment and analysis that was developed in 1976.18 However, it does not differentiate between a pure oculomotor deficit and a problem of random automated naming (RAN). Thus, a subsequent test was developed in 199019 and used in the assessment of general oculomotor problems, especially as related to reading, namely the Developmental Eye Movement (DEM) test. It is similar to the K-D test, but it circumvents this potential RAN problem.
The present pilot investigation sought to determine if the DEM test could be used in both the adult concussion/mild traumatic brain injury (mTBI) population, as well as the adult stroke or cerebral vascular accident (CVA) population, with an emphasis on quantifying the effects of controlled, laboratory-performed, oculomotor-based vision therapy/vision rehabilitation using the DEM test as a primary clinical outcome measure.
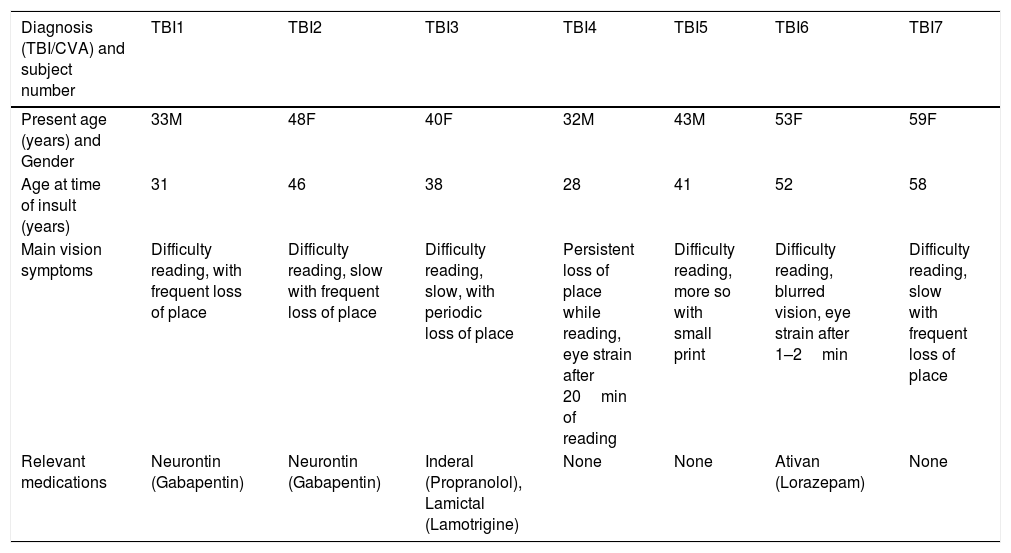
MethodsSubjectsThe subjects were comprised of fourteen adults who were medically diagnosed with acquired brain injury (ABI). Their clinical information is outlined in Table 1. Ages ranged from 30 to 73 years, with nine having mTBI and five CVA. They manifested various abnormal oculomotor signs, such as saccadic dysmetria and increased saccadic latency. Presenting relevant symptoms included reduced reading speed and impaired accuracy when reading. These signs and symptoms are consistent with the versional, oculomotor-based component of their reading dysfunction.20–23
Clinical data regarding subjects, nine of whom had TBI and five of whom had CVA.
| Diagnosis (TBI/CVA) and subject number | TBI1 | TBI2 | TBI3 | TBI4 | TBI5 | TBI6 | TBI7 |
|---|---|---|---|---|---|---|---|
| Present age (years) and Gender | 33M | 48F | 40F | 32M | 43M | 53F | 59F |
| Age at time of insult (years) | 31 | 46 | 38 | 28 | 41 | 52 | 58 |
| Main vision symptoms | Difficulty reading, with frequent loss of place | Difficulty reading, slow with frequent loss of place | Difficulty reading, slow, with periodic loss of place | Persistent loss of place while reading, eye strain after 20min of reading | Difficulty reading, more so with small print | Difficulty reading, blurred vision, eye strain after 1–2min | Difficulty reading, slow with frequent loss of place |
| Relevant medications | Neurontin (Gabapentin) | Neurontin (Gabapentin) | Inderal (Propranolol), Lamictal (Lamotrigine) | None | None | Ativan (Lorazepam) | None |
| Diagnosis (TBI/CVA) and subject number | TBI8 | TBI9 | CVA1 | CVA2 | CVA3 | CVA4 | CVA5 |
|---|---|---|---|---|---|---|---|
| Present age (years) and gender | 41M | 30F | 48M | 51M | 53M | 73M | 73M |
| Age at time of insult (years) | 39 | 29 | 47 | 50 | 40, 53 | 72 | 72 |
| Main vision symptoms | Difficulty reading, words “dance” on the page | Slow and labored reading, sensitivity to visual motion, intermittent double vision | Difficulty reading, slow, with frequent loss of place, right hemifield defect | Significant difficulty reading with frequent loss of place, right hemifield defect | Difficulty reading, blurred vision, left hemifield defect with inattention | Slowed speed and accuracy of reading, left hemifield defect with inattention | Difficulty reading, with frequent loss of place, left hemifield defect with inattention |
| Relevant medications | Neurontin (Gabapentin) | Klonopin (Clonazepam), Depakene (Valproic acid) | None | Coumadin (Warfarin) | None | Xalatan (Latanoprost), Timoptic (Timolol maleate) | Diovan (Valsartan), Celexa (Citalopram hydrobromide) |
TBI, traumatic brain injury; CVA, cerebral vascular accident; M, male; F, female.
All five stroke subjects were male, whereas 4 of the 9 mTBI subjects were male. Best-corrected visual acuity at distance and near for each subject was at least 20/30 in each eye. There was no manifest strabismus, nor any history of prior TBI, CVA, or neurological disease. Subjects presented with stable general health and sufficient cognitive ability to participate fully in and complete the eleven-week study. Regardless of the medications taken by the subjects,24 none reported blur or diplopia during the study.
All subjects were referred from either local hospitals or rehabilitative centers at least one year following their brain insult, and this ensured that changes during training would not be secondary to natural neurological recovery.25 Consequently, subjects presented to the Raymond J. Greenwald Rehabilitation Center at the SUNY-College of Optometry's University Eye Center with complete medical documentation, including information regarding acute and chronic care, neuro-imaging data, neurological reports, trauma history, and rehabilitative history. The research was approved by the college's IRB Committee, and it followed the tenets of the Declaration of Helsinki. Written informed consent was obtained from each subject.
The subjects received a detailed neuro-optometric rehabilitation (NOR) examination in the Raymond J. Greenwald Rehabilitation Center prior to participating in the study.26 This NOR vision examination included best-corrected monocular and binocular visual acuity (at distance and near), refractive state (at distance and near), ocular alignment and vergence ranges (at distance and near), near point of convergence, random-dot stereopsis at near, accommodative near testing (lag, amplitude, and dynamic facility for subjects under 40 years of age), DEM testing, monocular color vision, and ocular health (including biomicroscopy, applanation tonometry, dilated fundus examination, and automated visual fields).27,28
The Developmental Eye Movement (DEM) testThe DEM test19 (see Fig. 1) is comprised of one pre-test card and three test cards. The pre-test card is designed to ensure that the patient understands the test and can recognize, as well as consistently name, single digit numbers ranging from 1 to 9. The remaining three cards comprise the test. The test is typically performed at near (40cm) under binocular-viewing conditions with the patient's habitual near vision correction in place.
Test cards A and B assess the global efficiency of vertical saccades. Each card is comprised of two columns of evenly-spaced numbers. Without using one's finger, the patient is instructed to state the numbers aloud as accurately and quickly as possible from the top to bottom of one column, and the same with the second column. This is repeated for Test Card B. The combined time and errors resulting when stating the numbers for Test Cards A and B are noted as the vertical time and vertical errors, respectively.
Test Card C assesses the global efficiency of horizontal saccades. It is comprised of the same numbers in the same order as in Test Cards A and B, except that the numbers are oriented horizontally and now unevenly spaced. Without using one's finger, the patient is instructed to state the numbers aloud as correctly and quickly as possible from left to right, going down row by row, as if reading a text in English. The time and errors resulting when stating the numbers for Test Card C are noted as the horizontal time and horizontal errors, respectively.
Scoring of the test involves noting errors made for the three test cards, as well as comparing the time to call out the numbers horizontally to the time to call out the exact same numbers in the same order vertically. If the ratio for the time of calling out numbers horizontally relative to vertically is equal to 1.0, there is no clinical global horizontal saccadic deficiency. However, if both the vertical and horizontal times are longer relative to the normative data, then it is suggestive of a RAN, rather than a global saccadic problem per se. If the ratio for time of calling out the numbers horizontally relative to vertically is greater than 1.0, this suggests a clinical global horizontal saccadic deficiency.
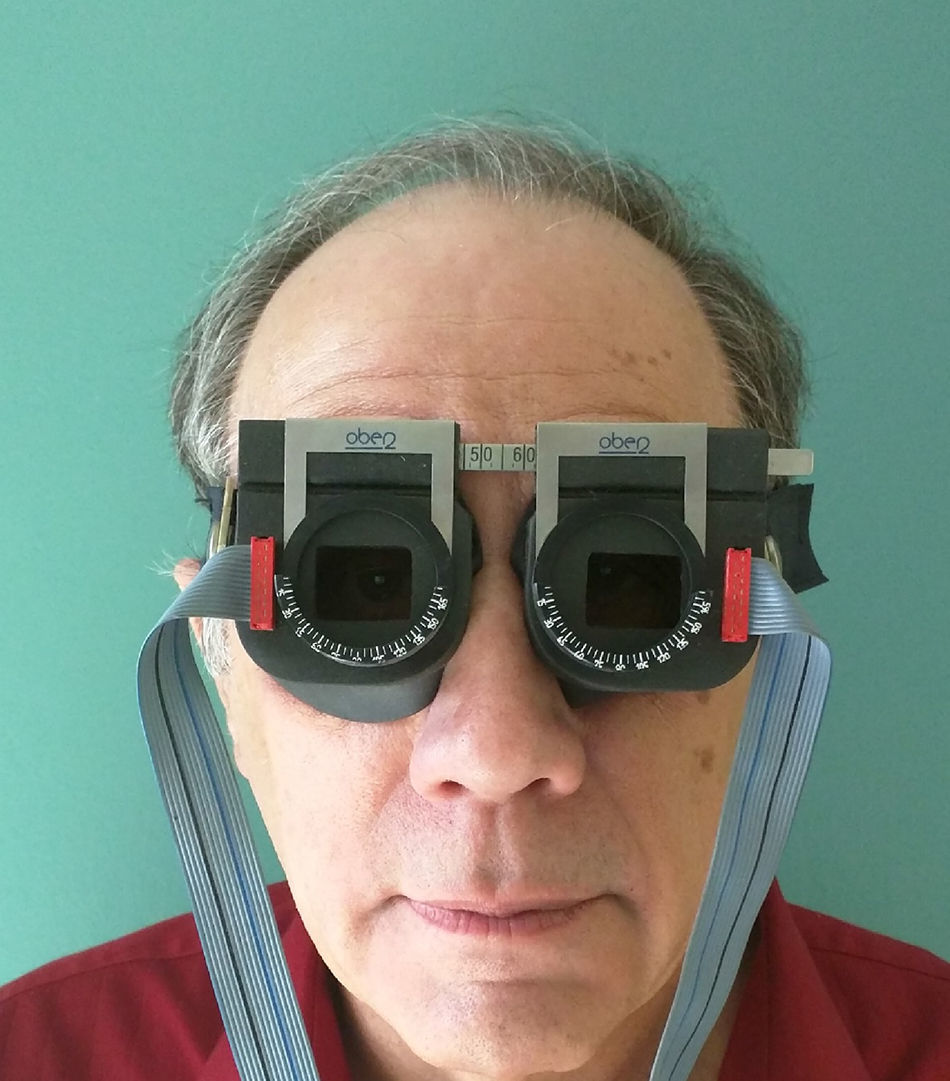
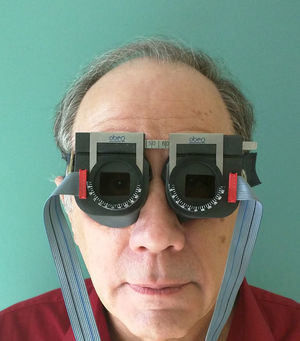
ApparatusHorizontal and vertical versional (i.e., conjugate) eye movements (i.e., fixation, saccades, simulated reading, and smooth pursuit) were assessed under binocular-viewing conditions. They were recorded objectively (see Fig. 2) using the OBER2 recording system (Permobil/IOTA Eye Trace, Sundsvall, Sweden).29 Auditory oculomotor feedback software and hardware related to eye position were custom-designed and integrated into the OBER2 system. This system incorporated goggle-mounted, infrared limbal reflection eye movement circuitry, with an ocular resolution of approximately 0.25 degrees, bandwidth dc to 120Hz, and a linear range of ±20 degrees both horizontally and vertically. The eye movement signals and stimulus timing indicators were input on-line to the computer, while the data analysis was performed manually off-line. The computer-controlled test stimuli consisted of a single, 30min arc, bright square viewed on a computer display monitor at a 40cm test distance. It was presented binocularly to test and train fixation, saccades, simulated reading, and pursuit.
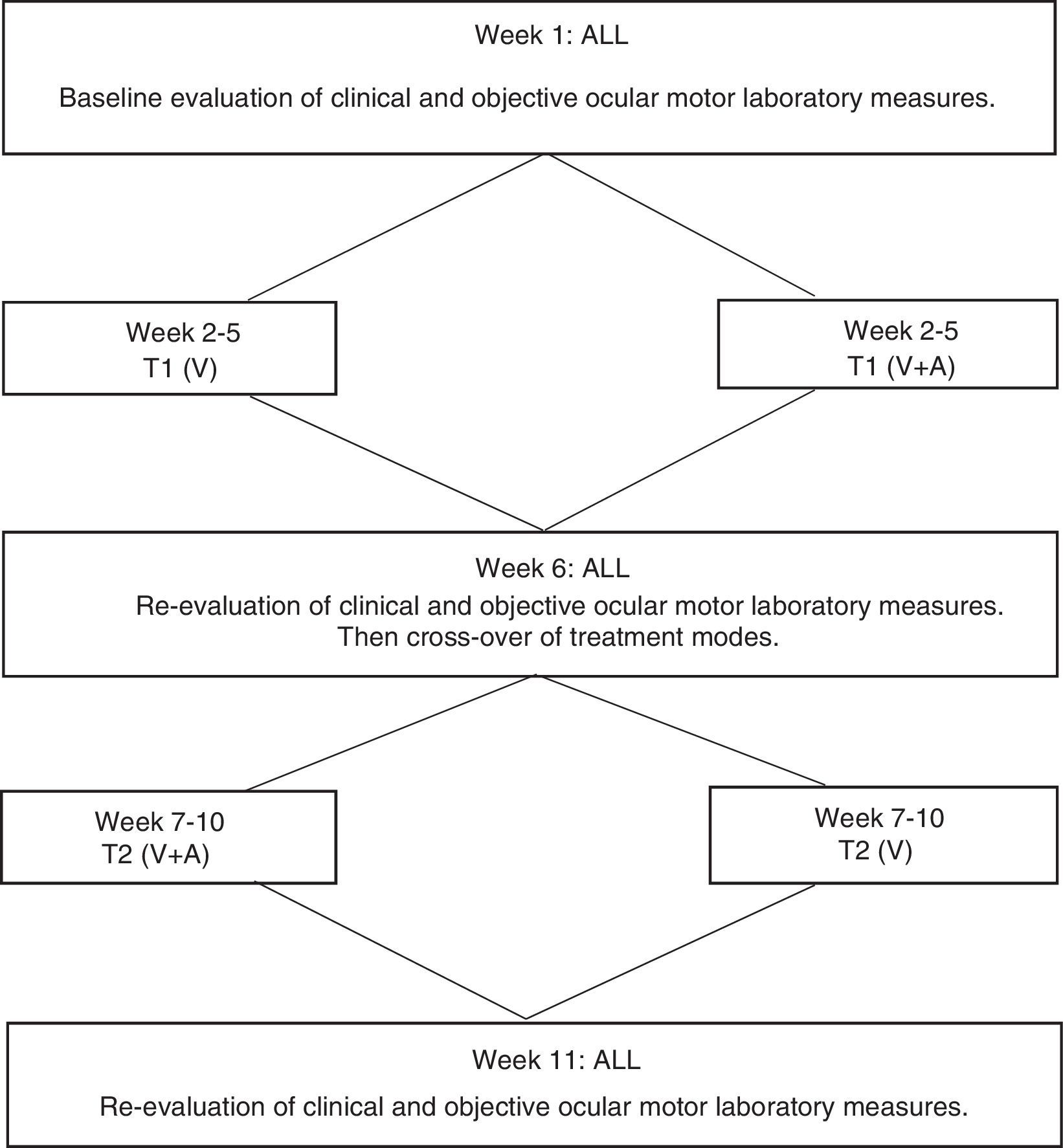
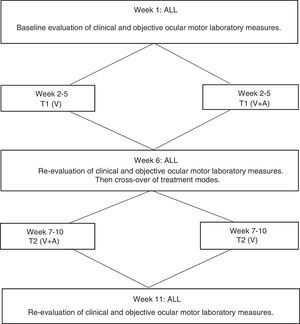
Study designThis study involved a consecutive 11-week commitment by each subject. The study design (see Fig. 3) was developed by the authors and described in detail elsewhere.20–22 It had five primary components:
- (1)
The initial clinical assessment and oculomotor laboratory evaluation were performed during week 1.
- (2)
The training portion commenced during week 2. The subject received 2 training sessions per week; each session was 60min in duration, involving 36min of actual training per session, with the remainder of time consisting of frequent, short, interspersed rest periods for the subject, for weeks 2–5 for the first phase of training (referred to as T1).
- (3)
During week 6, the subject was re-evaluated, including DEM testing, as in week 1.
- (4)
The subject was again seen twice weekly during weeks 7–10 for the second phase of training (referred to as T2).
- (5)
In week 11, the subject was re-evaluated, including DEM testing, as in weeks 1 and 6.
An interventional, cross-over experimental design (see Fig. 3) was employed, with each subject receiving the two feedback training modes (T1 and T2)20,22,30: (1) oculomotor rehabilitation employing one's normal internal visual feedback only (T1), and (2) oculomotor rehabilitation employing one's normal internal visual feedback concurrent with laboratory-based external auditory oculomotor feedback (T2), that is, subjects could also now “hear” their eye movements. Subjects were assigned to one of the two training modes (see Fig. 3) in a counter-balanced manner separately for each of the two diagnostic sub-groups. That is, if a subject were initially assigned to visual feedback (V) only for the first phase of training (T1), that subject was crossed over to receive the combined visual and auditory (V+A) feedback for the second phase (T2) of training, and vice versa for the next subject, in that particular diagnostic sub-group (i.e., either TBI or CVA). Thus, all training, as well as subsequent evaluations and analyses, were performed both separately, as well as combined, for the TBI and CVA diagnostic sub-groups to compare training effects.
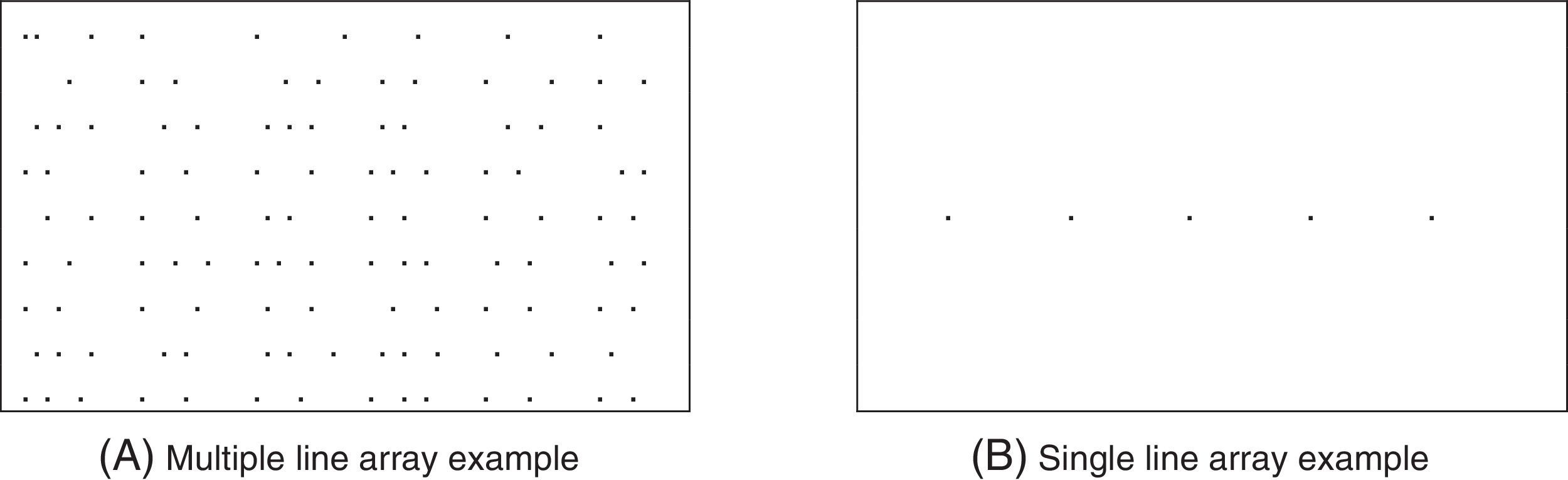
Test proceduresThe objective test procedures for versional eye movement assessment were developed by the authors and are described in detail elsewhere.20,22 Briefly, fixation, saccade, simulated reading, and pursuit eye movements were tested objectively under binocular-viewing conditions. Subjects were instructed to fixate, and follow, the center of the luminous square target20 (See Table 2 for the specific test conditions used and Fig. 4 for an illustration of the simulated reading stimuli). These basic, objectively-based versional eye movement findings will be the topic of a separate report, and they are included here only for completeness of describing the entire study protocol. Thus, the present paper will be restricted to reporting solely on the DEM results pre-, mid-, and post-training.
Oculomotor testing protocol sequence. Reprinted with permission from Table 1 on page 5 of Brain Research Protocols. 2004; Volume 14.
| Stimulus | Test duration (min) |
|---|---|
| 1.Central fixation | 0.5 |
| 2.Horizontal predictable saccades (+/− 10 deg amplitude, 0.33 cycle/s step intervals) | 0.5 |
| 3.Vertical predictable saccades (+/− 10 deg amplitude, 0.33 cycle/s step intervals) | 0.5 |
| 4.Horizontal random saccades (0.5–10 deg amplitudes, 0.3–3.8s step intervals) | 0.5 |
| 5.Vertical random saccades (0.5–10 deg amplitudes, 0.3–3.8s step intervals) | 0.5 |
| 6.Horizontal pursuit (+/− 10 deg amplitudes, 4.1 deg/s velocity) | 1.0 |
| 7.Vertical pursuit (+/− 10 deg amplitudes, 5.4 deg/s velocity) | 1.0 |
| 8.Multiple-line simulated reading (full page, 34 deg centered field, 1–3 deg amplitudes, 1.0 cycle/s step intervals) | 2.0 |
| 9.Single-line simulated reading (12 deg centered field, 3 deg step displacements, 0.33 cycle/s step intervals) | 1.0 |
| 10.Multiple-line silent reading (Visagraph, level 3, 2 selections) | Approximately 7.5 |
| 11.Multiple-line silent reading (Visagraph, level 10, 2 selections) | Approximately 7.5 |
| Total testing time | Approximately 22.5 |
The tasks in the two training modes were similar to that described earlier for the evaluation of fixation, saccades, simulated reading, and pursuit,20,22 except for the duration of the stimulus presentation (see Table 3). The difference in training was that the V mode incorporated only the subject's normal internal visual oculomotor feedback, while the V+A mode incorporated this normal internal visual feedback and the external auditory oculomotor feedback concurrently. The auditory oculomotor feedback for the V+A condition was incorporated by adding an auditory tonal change that was related (i.e., correlated) either with the subject's horizontal or vertical eye position, with each assessed separately.30–32 The auditory tone generator had an eye position-to-tone resolution of approximately 0.25 degrees both horizontally and vertically, with a tonal frequency range from approximately 2000 to 5000Hz. The subject received prior verbal description of the tonal expectations for optimal tracking of each stimulus.
Oculomotor training protocol sequence. Reprinted with permission from Table 2 on page 5 of Brain Research Protocols. 2004; Volume 14.
| Stimulus | Test duration (min) |
|---|---|
| 1.Central fixation (0 deg, midline) | 1.5 |
| 2.Horizontal predictable saccades (+/− 5 deg amplitude, 0.33 cycle/s step intervals) | 2.0 |
| 3.Horizontal pursuit (+/− 10 deg amplitude, 4.1 deg/s velocity) | 1.5 |
| 4.Multiple-line simulated reading (full page, 34 deg centered field) | 2.0 |
| TRAINING TIME SUBTOTAL=7.0min | |
| 5.Leftward fixation (10 deg left of midline) | 1.5 |
| 6.Horizontal predictable saccades (+/− 10 deg amplitude, 0.33 cycles/s step intervals) | 2.0 |
| 7.Horizontal pursuit (+/− 10 deg amplitude, 4.1 deg/s velocity) | 1.5 |
| 8.Single-line simulated reading (13 deg centered field) | 1.0 |
| TRAINING TIME SUBTOTAL=6.0min | |
| 9.Rightward fixation (10 deg right of midline) | 1.5 |
| 10.Vertical predictable saccades (+/− 5 deg amplitude, 0.33 cycle/s step intervals) | 2.0 |
| 11.Vertical pursuit (+/− 5 deg amplitude, 4.1 deg/s velocity) | 1.5 |
| 12.Multiple-line simulated reading (full page, 34 deg centered field) | 2.0 |
| TRAINING TIME SUBTOTAL=7.0min | |
| 13.Upward fixation (10 deg above midline) | 1.5 |
| 14.Vertical predictable saccades (+/− 10 deg amplitude, 0.33 cycles/s step intervals) | 2.0 |
| 15.Vertical pursuit (+/− 10 deg amplitude, 4.1 deg/s velocity) | 1.5 |
| 16.Single-line simulated reading (13 deg centered field) | 1.0 |
| TRAINING TIME SUBTOTAL=6.0min | |
| 17.Downward fixation (10 deg below midline) | 1.5 |
| 18.Horizontal non-predictable saccades (0.5–10 deg amplitudes, 0.3–3.8 s step intervals) | 2.0 |
| 19.Multiple-line simulated reading (full page, 34 deg centered field) | 2.0 |
| TRAINING TIME SUBTOTAL=5.5min | |
| 20.Central fixation (0 deg, midline) | 1.5 |
| 21.Vertical, non-predictable saccades (0.5–10 deg amplitudes, 0.3–3.8 s step intervals) | 2.0 |
| 22.Single-line simulated reading (13 deg field, centered on midline) | 1.0 |
| TRAINING TIME SUBTOTAL=4.5min | |
| TOTAL TRAINING TIME=36.0min | |
The total training time per session was 36min. For the visual feedback mode (V), subjects were instructed to track the target as accurately as possible with their eyes only, that is, without any extraneous head or body movements. They did not wear the eye movement system for the V training mode. However, for the combined visual and auditory feedback mode (V+A), subjects did wear the eye movement recording goggle system, as a tone related to eye position was present at all times during visual tracking of the specified target, again without any extraneous head or body movements. The training components are described below:
- (1)
For fixation, subjects in the V mode were instructed to maintain their eyes on the target as steadily as possible. Subjects in the V+A mode were additionally instructed to maintain the tonal pitch constant, per the verbal description: the steadier the tone, the steadier the fixational eye movements.
- (2)
For saccades, subjects in the V mode were instructed to move their eyes rapidly, accurately, and discretely as the target changed position. Subjects in the V+A mode were additionally instructed to generate a single, discrete, and rapid change in tonal pitch, per the verbal description; such a tonal change would be created by a single accurate saccade, which would be ideal. Any undesirable subsequent corrective saccades would be heard as additional discrete and rapid tonal changes, but with less variation in pitch, as these would typically be smaller in amplitude.
- (3)
For pursuit, subjects in the V mode were instructed to follow the moving target as accurately as possible. Subjects in the V+A mode were additionally instructed to create a smooth and continuous change in tonal pitch, per the verbal description; such a tonal change would be created by optimal saccade-free smooth pursuit. The presence of saccades within (i.e., superimposed upon) the pursuit movement would create discrete pitch changes interspersed within the smooth and continuous tonal change.
- (4)
For simulated reading, the instructions and goals were the same as that for basic saccadic tracking. Subjects in the V mode were instructed to move their eyes rapidly, accurately, and discretely as the target changed position. Subjects in the V+A mode were additionally instructed to generate a single and discrete tonal change for each discrete step displacement of the target.
The following statistical analyses were performed using Excel Analysis ToolPak, version Excel 2010. Due to the small sample size, descriptive and non-parametric (binomial test) statistical analyses were performed in most cases. The binomial test compares the number of subjects in a group that changed (or improved in this case) versus the total number of subjects in the group, with the resultant probability. The t-test was also used as indicated in the text below. A p-value of 0.05 or less was considered to be statistically significant.
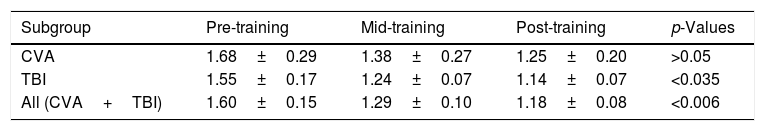
ResultsDEM ratioThe DEM ratio was compared for the stroke subgroup (n=5), TBI subgroup (n=9), and the entire sample (n=14) across the training periods: pre-, mid-, and post-training (see Table 4A).
Individually, the DEM ratio for four out of the five subjects with stroke decreased numerically with training, thus suggestive of improvement. However, it was not statistically significant based upon the binomial test (p>0.05).
Individually, the DEM ratio for eight out of nine subjects with TBI decreased with training. This was statistically significant based upon the binomial test (p<0.035).
Individually, the DEM ratio for four of the five stroke subjects, and eight of the nine subjects with TBI, decreased with training (12 out of 14). This was statistically significant based upon the binomial test (p<0.006).
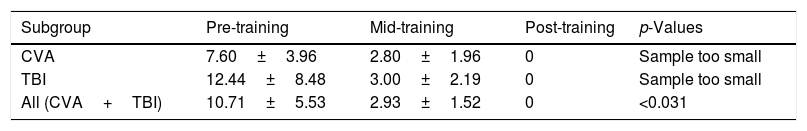
DEM errorsThe DEM errors were compared for the stroke subgroup (n=5), TBI subgroup (n=9), and the entire sample (n=14) between two training periods only: namely, pre- and mid-training, since all subjects manifested no errors at the post-training assessment (see Table 4B).
For the DEM errors in those with stroke, only three of the five manifested errors at the pre-training level. All three (100%) showed a considerable reduction in the number of errors at the mid-training level.
For the DEM errors in those with TBI, only three of the nine manifested errors at the pre-training level. All three (100%) showed a considerable reduction in the number of errors at the mid-training level.
For DEM errors in the entire sample (n=14), only six subjects manifested errors at the pre-training level. All six (100%) showed a reduction in the number of errors at the mid-training level, which was statistically significant based on the binomial test (p<0.031).
Effect of visual only versus visual plus auditory feedbackThe effect of V only versus V+A feedback training was also assessed. There was a trend (t-test, p<0.10) for improved oculomotor control with the combined V+A feedback training mode in both groups.
DiscussionThe findings of the present pilot investigation are promising. This is the first study to demonstrate efficacy of the DEM test before and after oculomotor-based vision rehabilitation in both the adult mTBI and stroke population subgroups of ABI. Significant therapeutic effects were found in several instances, even in the small sample sizes tested and relatively short period of training, which suggests robustness of the intervention and resultant effect. Since many of these individuals with ABI have short- and long-term memory problems, it is not likely that these positive training effects represent a ‘learning effect’, given that the test periods were separated by about 6 weeks.
Both the present, and related earlier findings in these populations,5,7,20–23,33,34 demonstrate the considerable degree of residual visual system/brain plasticity, even in the adult, neurologically-compromised brain. Furthermore, persistent, positive therapeutic effects have been demonstrated both subjectively in the clinic23 and objectively in the laboratory, both in the short5,7,20–23,33,34 and long-term,33 along with reduced visual symptoms,23 improved visual/general attention 35,36, and improved reading ability (e.g., reading longer without visual symptoms).23
The present positive findings suggest that one should consider using the DEM test in a wide variety of contexts, in addition to evaluating the effect of some visual intervention, as was conducted in the present investigation. For example, it could be used in both the sports arena and in the military to establish a normative baseline prior to the playing season or deployment, respectively. These baseline DEM findings could be then used to assess for the presence of a possible concussion in the following weeks and months. The DEM test could also be used in the emergency room to assess for a range of head injuries that might impair saccadic eye movement ability and related visual attention; this would be done in addition to the conventional armamentarium of tests, such as the pupillary light reflex. Moreover, the DEM could be used to assess for positive changes in these patients as they pass through the subacute and then into the chronic phases of their natural recovery over the subsequent several months. And, if the DEM test proves to be a good adjunct test in the acute diagnosis of concussion/mTBI, then the clinician would now have two similar tests to use and compare: namely, the DEM and the K-D tests. Lastly, the DEM test may serve in a vision screening capacity in an ABI clinical practice.37
While studies in TBI have centered on those with the ‘mild’ variety, which constitutes the majority of cases,38 the DEM test is rapid, simple, and portable, and, hence it could be used in the clinic or bedside in both the moderate and severe TBI populations, respectively, namely in those that do not have markedly impaired speech, linguistic, and/or cognitive aspects. It could be used to assess their baseline level upon clinical or hospital entry, and then later employed to assess and quantify the effects of either the natural recovery process or some vision intervention, such as vision therapy or prisms.39
Limitations and future directionsThere were some study limitations and some future directions for research. First, as it was a pilot investigation, the sample sizes were by nature small in each subgroup. Future investigations should include a randomized clinical trial with much larger sample sizes as determined formally by a statistical power analysis. Second, the study did not include either a control or placebo group. Third, the actual total duration of training over the 11-week period was only 9.6h, which in the clinical domain is short for the ABI population. Typically, a range of vision rehabilitation procedures (primarily oculomotor-based) might take 20–30h, or more, to attain a stable, high level of performance.5,33,34,39 Fourth, and to our surprise, several subjects in each group failed to exhibit any errors in the DEM test at baseline. This made comparisons over the training period more tenuous. A future investigation should include a comprehensive baseline analysis with more subjects in each subgroup across various ages of adults to ascertain if there was a sampling bias (e.g., convenience sampling) and investigate any effect of age in an adult population. Fifth, and last, the oculomotor training only included the versional system. It did not include the vergence and accommodative systems, and their mutual interactions between themselves and also with the versional system, as has been done more recently in the first clinical trial of its type in the adult mTBI population,5,7,23,33,40 in which an even greater positive effect might be anticipated.
ConclusionsThe positive results of the present study show promise in the future for diagnostic, prognostic, and therapeutic assessment in the ABI population. Such investigations should be conducted, perhaps at the acute, subacute, and chronic stages of both mTBI and stroke to determine if there are substantial differences that must be taken into account as they progress after their initial brain insult. Moreover, the DEM findings can be used in conjunction with other clinical results, such as general attention/visual attention testing/training, and cognitive testing/training involving visual saccadic scanning, to gain a better and more comprehensive sense of the patient's visual and overall brain's functional capabilities.
Conflicts of interestThe authors have no conflicts of interest to declare.
The authors thank the Langeloth Foundation for support of this research. The authors thank Dr. Jan Ygge for the loan of the eye movement software programs.