To investigate non-cycloplegic changes in refractive error prior to the onset of presbyopia.
MethodsThe Aston Longitudinal Assessment of Presbyopia (ALAP) study is a prospective 2.5 year longitudinal study, measuring objective refractive error using a binocular open-field WAM-5500 autorefractor at 6-month intervals in participants aged between 33 and 45 years.
ResultsFrom the 58 participants recruited, 51 participants (88%) completed the final visit. At baseline, 21 participants were myopic (MSE -3.25±2.28 DS; baseline age 38.6±3.1 years) and 30 were emmetropic (MSE −0.17±0.32 DS; baseline age 39.0±2.9 years). After 2.5 years, 10% of the myopic group experienced a hypermetropic shift (≥0.50 D), 5% a myopic shift (≥0.50 D) and 85% had no significant change in refraction (<0.50 D). From the emmetropic group, 10% experienced a hypermetropic shift (≥0.50 D), 3% a myopic shift (≥0.50 D) and 87% had no significant change in refraction (<0.50 D). In terms of astigmatism vectors, other than J45 (p<0.001), all measures remained invariant over the study period.
ConclusionThe incidence of a myopic shift in refraction during incipient presbyopia does not appear to be as large as previously indicated by retrospective research. The changes in axis indicate ocular astigmatism tends towards the against-the-rule direction with age. The structural origin(s) of the reported myopic shift in refraction during incipient presbyopia warrants further investigation.
Investigar los cambios en el error refractivo sin cicloplégico con anterioridad a la aparición de la presbicia.
MétodosEl estudio de la evaluación longitudinal de la presbicia de Aston (ALAP) es un estudio longitudinal prospectivo de 2,5 años que mide el error refractivo objetivo utilizando un autorrefractor binocular de campo abierto WAM-5500 a intervalos de 6 meses, en participantes con edades comprendidas entre 33 y 45 años.
ResultadosDe los 58 participantes estudiados, 51 de ellos (88%) completaron la visita final. Al inicio, 21 participantes eran miopes (MSE -3,25±2,28 DS; edad basal: 38,6±3,1 años) y 30 eran emétropes (MSE -0,17±0,32 DS; edad basal: 39±2,9 años). Transcurridos 2,5 años, el 10% del grupo de participantes miopes experimentó un cambio hipermetrópico (≥0,5 D), el 5% un cambio miópico (≥0,5 D), y el 85% no experimentó cambio refractivo significativo alguno (<0,5 D). En el grupo emétrope, el 10% experimentó un cambio hipermetrópico (≥0,5 D), el 3% un cambio miópico (≥0,5 D), y el 87% no experimentó cambio refractivo significativo alguno (<0,5 D). En términos de vectores astigmáticos, todas las mediciones permanecieron invariables durante el periodo de estudio excepto J45 (p<0,001).
ConclusiónLa incidencia del cambio miópico en la refracción durante la presbicia incipiente no parece ser tan grande como anteriormente indicado en investigaciones retrospectivas. Los cambios en los ejes indican que el astigmatismo ocular tiende hacia la dirección contra la norma con la edad. El(los) origen(es) estructural(es) del cambio miópico reportado en la refracción durante la presbicia incipiente justifica la investigación futura.
Age-related changes in refraction have been well documented cross-sectionally and longitudinally during childhood,1,2 early adulthood3,4 and presbyopia.5,6 Whilst a significant body of work charts myopia genesis and progression in children,7–13 there is also evidence of myopia onset and progression during adulthood.4,14,15 Indeed, Rahi et al.16 reported 49% of 2487 randomly selected British adults aged 44 years were myopic (MSE≤−0.75 D), with over 80% of the myopia occurring after the age of 15 years (late onset myopia).17,18
Considering the typical change in refraction between the ages of 35–65 years,19 a hypermetropic shift has consistently been documented by cross-sectional14,20–29 and longitudinal5,6,15 studies, with a myopic shift reported after 65 years, possibly due to the onset of crystalline lens nuclear sclerosis.30 Current understanding of the optical and structural changes that occur during the development of presbyopia suggest the origin of the hypermetropic shift between the ages of 35–65 years could be the manifestation of previously latent hypermetropia,31 which can no longer be overcome due to a reduction in amplitude of accommodation, or the crystalline lens paradox, where the increase in crystalline lens thickness and curvature is over-compensated for by a reduction in the average refractive index of the crystalline lens.5,14,32
However, Pointer and Gilmartin33 have presented retrospective data revealing 20% of myopic participants experienced a myopic shift in refraction of 0.50–0.75 D between the ages of 35–44 years, otherwise classified as the period of incipient presbyopia i.e. before a reading addition correction is considered clinically necessary. Incipient presbyopic participants were largely omitted from the aforementioned adult studies. Despite reporting a hypermetropic shift (≥0.50 D) in refraction in emmetropic and hypermetropic individuals after the age of 40 years, Grosvenor and Skeates5 isolated retrospective longitudinal myopic participant data to find the hypermetropic shift in refraction was less prevalent amongst myopic participants (19%). In fact, most myopic participants remained stable (66%) or became more myopic by ≥0.50 D (15%) after the age of 40 years. Further, refractive data from 15 population-based cohort and cross-sectional studies analysed by the European Eye Epidemiology (E3) Consortium demonstrates a small (1.7%) increase in the prevalence of low myopia (classified as ≤−0.75 to >−3.00D) between the ages of 35–39 years and 40–44 years, which could reflect further a myopic shift in the incipient phase of presbyopia.19 The ocular changes driving this putative myopic shift in refraction during incipient presbyopia are currently unknown. Moreover, it is unclear why myopic individuals appear to be at a greater risk of a myopic shift in refraction than emmetropic individuals during incipient presbyopia. Perhaps the effects of the crystalline lens paradox are less pronounced in myopic eyes due to their thinner crystalline lenses4,17,34 or previous axial elongation acts as a predisposition for future continued axial elongation.
It is feasible that the putative myopic shift may act as a compensatory mechanism to overcome near vision blur resulting from diminishing levels of accommodation during incipient presbyopia. Indeed, a connection has been made between high levels of near work and myopia onset and progression in both children35,36 and adults.4,37–39 The myopic shift may, therefore, be more prevalent in individuals spending long periods of time working at near.
The aim of this study is to provide prospective longitudinal data documenting the natural progression of refraction during incipient presbyopia, and in particular to investigate the incidence of the putative myopic shift in refraction amongst myopic and emmetropic individuals. Refractive changes will also be considered with respect to the amount of time spent working at near daily to explore the link between near work and myopia during the incipient phase of presbyopia.
Materials and methodsIn order to collect longitudinal data, the study was designed to review participants every 6 months over 2.5 years, thus 6 sessions were completed in total, each time repeating the experimental protocol detailed below. One UK-registered optometrist (DL) collected the data at each visit.
The study was approved by the Aston University Audiology and Optometry Research Ethics Committee and was conducted in accordance with the tenets of the Declaration of Helsinki. Written informed consent was obtained from all the participants after an explanation of the nature and possible consequences of the study.
Sample sizeTo ensure the recruited sample size was appropriate for repeated measures ANOVA analysis (within and between interaction), an effect size (f) of 0.25, an error probability (α) of 0.05 and required power (1−β) of 0.80 was inputted into G*Power 340 for 6 repeat measurements amongst 2 groups, which produced an overall sample size of 20.
QuestionnaireIn order to confirm suitability to participate in the study, each volunteer was asked to complete a questionnaire. The questionnaire asked each participant to document their date of birth, ethnicity, date of last sight test, general health, medications and previous hospital eye service treatment. The British National Formulary41 was consulted to ensure none of the medications listed had any potential ocular adverse reactions that might affect the accommodative apparatus.
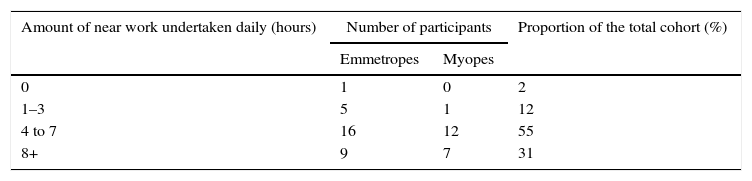
The questionnaire also collected information on the lifestyle and occupation of each participant. Due to the established links between high levels of near work and myopia progression in children both children35,36 and adults,4,37–39 participants were asked to indicate whether they spent 0, 1–3, 4–7 or 8+ hours working at near or using a computer daily. Low levels of outdoor sun exposure has also been linked with myopia,42 therefore each participant was asked whether their job was outdoor-based. Each participant was asked to recheck their completed questionnaire at each subsequent visit and were allowed to alter their responses as required (e.g. if their job had changed).
Visual acuityMonocular and binocular high contrast logMAR visual acuity was measured at 6m to ensure each participant could achieve an acuity of 0.1 logMAR or better. Near visual acuity was evaluated with a near logMAR card at 25cm to ensure each participant could achieve 0.1 logMAR or better at visit 1. At subsequent visits, visual acuity was re-measured to provide an indicator of changes in refraction, accommodation and the development of any pathology, but was not intended to be used as an outcome measure.
Amplitude of accommodationMonocular subjective amplitude of accommodation was measured in right eyes using an RAF rule (Richmond Products, New Mexico, USA) via the push-up/pull-down method.43,44 Whilst wearing their habitual refractive correction, participants were asked to wear an eye patch over their left eye and try to maintain clear focus of the high contrast N5 print as it was slowly pushed towards them until the point where they first reported sustained blur (push-up amplitude). Subsequently, the first point at which the N5 print became clear when moved away from the eyes (pull-down amplitude) was measured. If a participant was unable to achieve the minimum amplitude of accommodation measureable (2.00D), a +3.00D full aperture trial lens was introduced in front of their right eye. The power of the lens was subsequently subtracted from the push-up/pull-down amplitude value obtained. Push-up/pull-down amplitude was measured three times and averaged to calculate the mean amplitude of accommodation for the right eye.
Objective refractive error and keratometryObjective refractive error and keratometry were measured simultaneously by the binocular open-field WAM-5500 autorefractor (Grand Seiko Co. Ltd., Japan).45 Due to restrictions in room dimensions, a bespoke +5.00D Badal lens system with a high contrast Maltese cross target was mounted on the WAM-5500 autorefractor to enable distance (0D stimulus) viewing. The fixation target was placed at the focal length of the lens (20cm) in order to measure uncorrected distance refractive error. The left eye of each participant was occluded and participants were asked to focus on the centre of a Maltese cross as accurately as possible throughout data collection. Five consecutive measurements were acquired and the average MSE, J180, J45, cylinder (C), axis (θ) and power (P) were calculated.46 A refractive shift ≥0.50D in MSE was adopted as the minimum threshold level for statistical significance.47 The anterior corneal radii of curvature and toricity were also recorded. The radii of curvature readings from the principle meridians were averaged to determine the mean central keratometry reading.
Statistical analysisAll data from right eyes only were tested for normality using the Shapiro–Wilk test (SigmaPlot; Systat Software Inc., California, USA). The gender distribution within the emmetropic and myopic groups was compared using a Chi-Square Test. Differences between the baseline age of the emmetropic and myopic groups were determined by a t-test (SPSS; SPSS Inc., Illinois, USA). In order to determine whether changes in amplitude of accommodation and refraction vectors were significant over the 2.5 year period, repeated measures analysis of variance (ANOVA) testing was performed, comparing the effect of time (within-subject variable) and refractive group classification (between-subject variable). Linear regression analysis was performed to determine whether the baseline MSE, change in amplitude of accommodation and refraction vectors was significantly correlated with baseline age and whether the change in amplitude of accommodation was significantly correlated with the change in refraction after 30 months.
ResultsParticipantsIn order to allow for attrition during the course of the longitudinal study and to increase the likelihood of enrolling individuals who experience a myopic shift in refraction, 58 participants aged 33–45 years old (39.1±3.2 years) were recruited from an Aston University staff and local business volunteer call. All of the participants were screened to exclude those with a positive history of ocular or systemic disease. Each participant had visited an Optometrist for a full eye examination within 2 years from the baseline appointment and no one had been prescribed a reading addition.
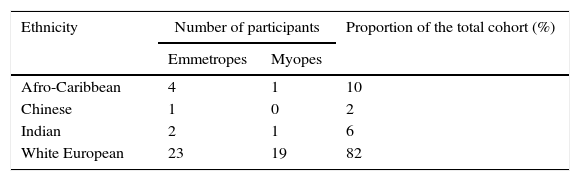
There was no attrition of subjects during the first year of the study, however, due to developed ocular (n=1) and systemic pathology (n=1) and permanent relocation from Birmingham (n=1), a total of 95% (n=55) of the original cohort completed the 2.5-year study visit. The cohort's ethnic composition is shown in Table 1. A total of 7% (n=4; mean baseline age 44.6±0.6 years) of participants required the +3.00D supplementary lens to measure their amplitude of accommodation. The same participants started wearing a near addition for reading during the course of the study and were, therefore, excluded from the analysis in this study. Of the remaining 51 participants, the average time between visits was 171±7 days. None of the participants who were pregnant during the study (n=4) reported gestational diabetes or any other health or visual issues.
Participants with a MSE of <−0.75 DS were defined as myopic.19 Emmetropes were classified by a MSE of between −0.75DS and +0.50DS. At baseline, 21 (14 females and 7 males) participants were myopic (MSE −3.25±2.28DS; baseline age 38.6±3.1years) and 30 (19 females and 11 males) were emmetropic (MSE −0.17±0.32DS; baseline age 39.0±2.9years). No restrictions were made based on the magnitude of cylindrical correction (myopes mean C −0.71±0.71DC; emmetropes mean C −0.39±0.32DC). The proportion of the myopic cohort who self-reported becoming myopic before the age of 15 years (early-onset myopes) was 57%. The amount of self-reported near work undertaken by the participants is reported in Table 2. Note, only 1 emmetropic individual reported spending most of the day outside. These figures did not change during the course of the study.
Cross-sectional analysisChi-square analysis revealed the male: female ratio within the myopic and emmetropic groups was not significantly different throughout the study (Chi-square=0.003, p=0.958). The baseline average age of the myopic (38.6±3.1 years) and emmetropic (39.0±2.9 years) groups were not statistically significantly different (t=0.463; p=0.646). Baseline MSE was not correlated with baseline age (r=0.263, p=0.062), whereas baseline right eye amplitude of accommodation was significantly correlated with baseline age (r=0.561, p<0.001).
Longitudinal analysisParticipants in the myopic group experienced a hypermetropic shift (≥0.50D; n=2, 10%), myopic shift (≥0.50D; n=1, 5%) or had no significant change in MSE (<0.50D; n=18, 85%). A similar proportion of the emmetropic group also experienced a hypermetropic shift (≥0.50D; n=3, 10%), myopic shift (≥0.50D; n=1, 3%) or had no significant change in MSE (<0.50D; n=26, 87%). The sphero-cylindrical refraction of the two myopic and emmetropic participants undergoing a myopic shift ≥0.50D changed from −5.72/−1.59×13 to −6.63/−1.21×13 and +0.15/−0.86×100 to −0.66/−0.85×101, respectively and corresponded with a drop in visual acuity.
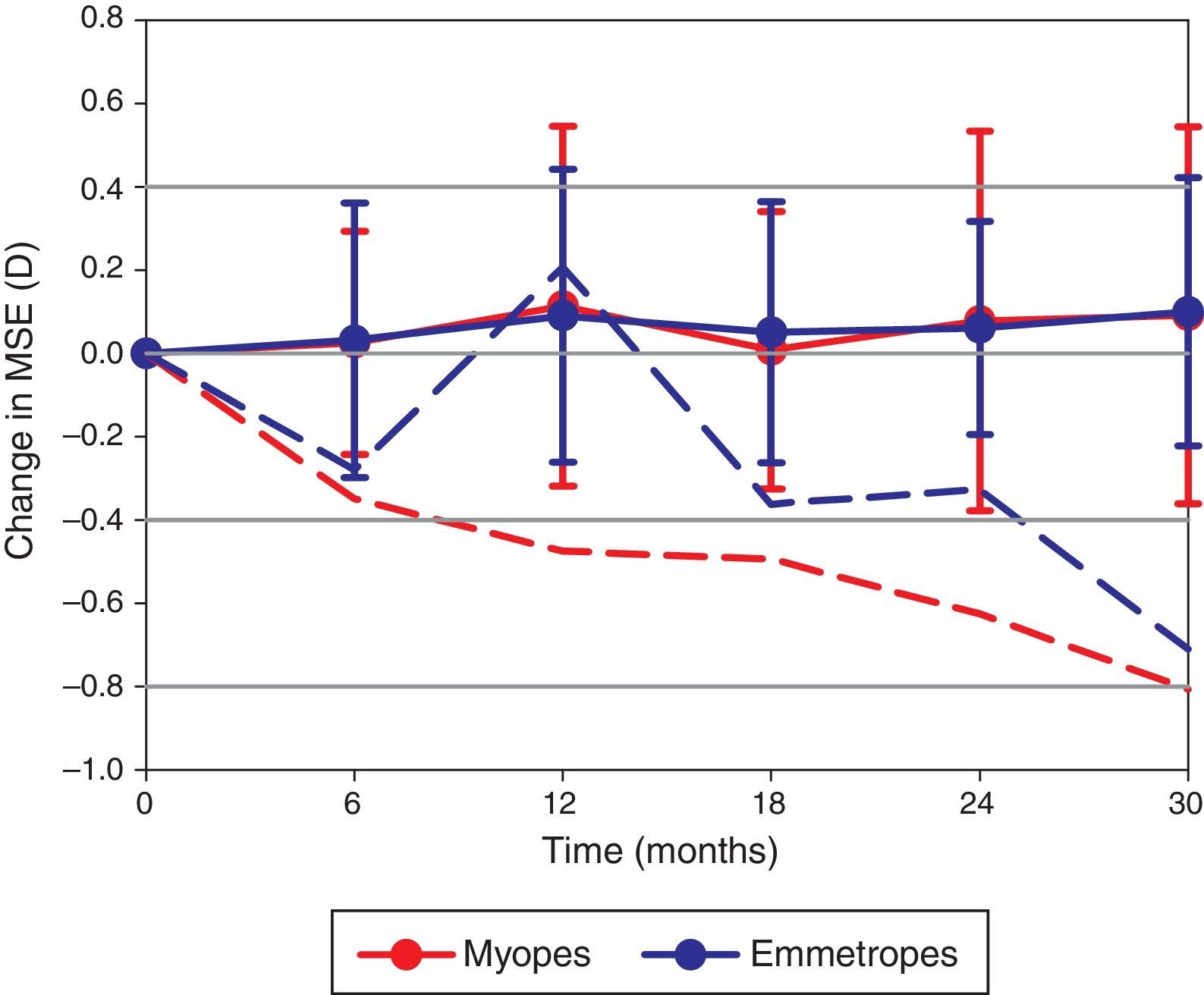
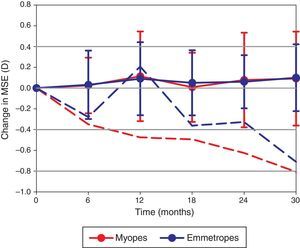
Three participants with myopia at baseline (MSE −1.19±0.35D) became emmetropic (MSE −0.39±0.23D) and one emmetropic participant (MSE −0.28D) became myopic (MSE −1.09D) during the course of the study. A repeated measures ANOVA test revealed the change in MSE was not significantly different between the myopic and emmetropic groups (F=0.150, p=0.980; Fig. 1). Furthermore, the overall change in MSE over the 2.5-year period (+0.10±0.38D) was not statistically significant (F=1.101, p=0.361). The rate of change in MSE was not correlated to baseline age (r=0.019, p=0.896).
The change in MSE documented at 6 visits over 2.5 years. The dashed lines represent the myopic (red) and emmetropic (blue) participants who became more myopic over the course of the study. The solid lines with error bars (±1 standard deviation) represent the refractive change observed in the remainder of the myopic and emmetropic cohort.
The overall change in astigmatism vector J180 was not found to be statistically significant (F=1.345, p=0.255). The change in J45 was statistically significant (F=8.085, p<0.001), changing from +0.02±0.19D at baseline to −0.06±0.19D at the final visit, with no interaction based on refractive error grouping emerging (F=0.736, p=0.597). Furthermore, the change in C (F=0.733, p=0.599), θ (F=0.887, p=0.491) and P (F=0.287, p=0.920) were found to be invariant over the course of the 2.5-year study. The average angle of the axis (θ) of astigmatism shifted from 156° at baseline to 144° at the conclusion of the study. The axis of the anterior corneal toricity shifted from 31° to 67°, which was statistically significant (F=6.047, p<0.001). The changes in the mean anterior corneal radius of curvature (F=0.834, p=0.527) and toricity (F=0.799, p=0.496) were not statistically significant.
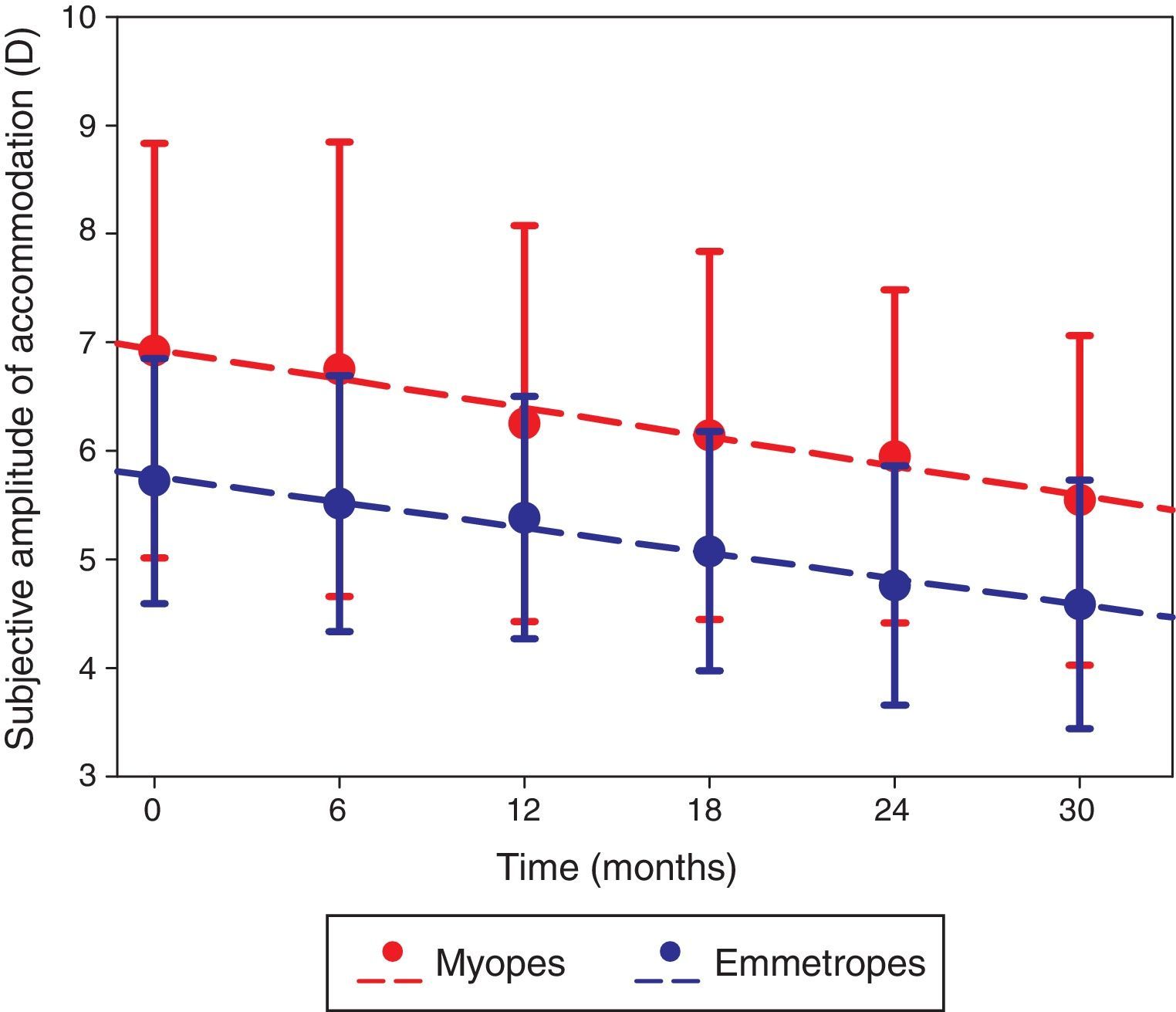
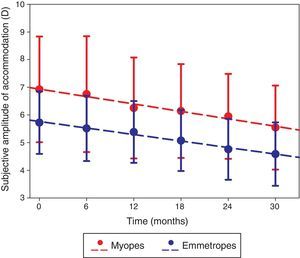
The amplitude of accommodation in the right eye significantly decreased over the 2.5-year study (F=37.219, p<0.001; Fig. 2), and was higher in the myopic group than the emmetropic group throughout the study (F=7.841, p=0.007). The rate of change over the 2.5-year study was not significantly different between the two refractive groups (F=1.213, p=0.307) and was not dependent on baseline age (r=0.045, p=0.752). The change in refractive error was not correlated to the change in amplitude of accommodation (r=0.028, p=0.847).
The right eye subjective amplitude of accommodation measured at each visit in myopic (red) and emmetropic (blue) participants with error bars (standard deviation). The dashed lines represent the regression line of the myopic (y=−0.269x+7.201; R2=0.969; p<0.001) and emmetropic (y=−0.236x+6.000; R2=0.986; p<0.001) data.
The present investigation represents the first prospective, longitudinal study to record changes in objective refraction during incipient presbyopia. Overall no significant change in refraction was observed, however a small proportion of participants experienced a significant hypermetropic or myopic shift in refraction (>0.50D).
Data from previous studies have indicated a hypermetropic shift in refraction would be expected within the age group recruited for this study,6,14 however, the average change in MSE over the course of the 2.5 year study, although hypermetropic, was not found to be statistically significant (p=0.242). A total of 4% (n=2) of the cohort underwent a myopic shift in refraction >0.50D over the 2.5 year study, which constituted 5% of the myopic and 3% of the emmetropic participants. Previous research has indicated between 15 and 20% of myopes and 3% of emmetropes experience a myopic shift in refraction >0.50D during incipient presbyopia.5,33 A hypermetropic shift ≥0.50D was also experienced in the myopic (10%) and emmetropic (10%) groups in the current study, with the remaining participants (85% of myopes and 87% of emmetropes) observing no significant change in MSE (<0.50 D). Grosvenor and Skeates5 reported 19% of myopic participants and 54% of emmetropic participants experienced a hypermetropic shift in MSE, whilst the MSE of the majority of myopic participants (66%) remained relatively stable (change <0.50D). Ellingsen et al.48 also found the MSE of the majority of myopes did not change ≥0.50D during incipient presbyopia, reporting shifts in MSE of −0.39±0.60D in 78 participants during their 30s and −0.29±0.56D in 130 participants during their 40s. The differences between refractive error progression of emmetropic and myopic individuals reported by previous studies have not been replicated by the current study, possibly as a result of selection bias inherent within the previous retrospective studies. However the data presented by Grosvenor and Skeates5 and Pointer and Gilmartin33 mapped refractive changes over a longer period (>4 years) and Grosvenor and Skeates’ study is likely to represent refractive changes during manifest presbyopia also.
A longitudinal study of similar length to the current study reported 45% of 322 eyes from 166 clinical microscopists aged 21 to 55 years experienced a myopic shift in MSE (≥0.375D, mean change −0.58±0.04D) in 2 years,4 however, data were not provided specifically on the pattern of refractive change according to age. Nevertheless, it is possible to ascertain from graphical representation of part of the data that 6 eyes belonging to individuals aged 40–42 years who were emmetropic (MSE between −0.25 to +0.625D) at the start of the study experienced a myopic shift in refraction (≥0.375D) and were classified as myopic (MSE≤−0.375D) at the end of the 2 year study. The proportion of myopic incipient presbyopes who underwent a myopic shift in refraction is unclear. Despite the suboptimal criteria for a significant change in refraction,47 the work of McBrien and Adams4 provides longitudinal evidence of a myopic shift within an occupational group who typically spending most of their day working at near. Furthermore, the participants who experienced a myopic shift in refraction in McBrien and Adams’ study were older (40–42 years) than the individuals who demonstrated a myopic shift in refraction in the present study (37 and 39 years), indicating the myopic shift also occurs in older incipient presbyopes than observed during the course of the current study.
The myopic participant (aged 37 years at baseline) who underwent a myopic shift in refraction of 0.71D over the 2.5 year study was diagnosed as myopic at age 4 (early-onset myopia) and entered the study with a MSE of −6.52D, self-reporting her refraction had stabilised during her adult years. It has been well-established that early-onset myopes are likely to progress to much higher levels of myopia than those who develop myopia after puberty.49,50 The trigger promoting continued myopia progression is unclear, however an increase in vitreous chamber depth has typically been identified as the structural correlate responsible.4,51 The emmetropic participant (aged 39 years at baseline) who underwent a myopic shift in refraction of 0.81D had no previous history of refractive error, although was of Chinese descent and gave birth 3 months after the 1 year review visit. The Chinese ethnicity has the highest prevalence of myopia of all ethnicities13,52,53 and hitherto the reason for this predisposition remains equivocal. Furthermore, pregnancy has been linked with myopic shifts in refraction, however the refractive error typically returns to pre-pregnancy levels post-partum.54 The small perturbation in the emmetropic participant's data at visit 3 is likely to be due to the data being from just one individual and, therefore, clinically insignificant noise. The participant from the current study underwent a further myopic shift of 0.33D from her third to sixth visit and was not diagnosed with gestational diabetes or any other complications, suggesting her refractive shift is unlikely to be pregnancy-related.
Considering ocular astigmatism during incipient presbyopia, the changes in the J180, C and θ presented here failed to reach statistical significance. Nevertheless, the change in J45 and the change in the axis of the mean cylinder from 156° to 144° indicate ocular astigmatism changes towards the against-the-rule direction (steeper horizontally) with age, which is agreement with previous studies.55–57 The axis of anterior corneal toricity also shifted in the against-the-rule direction from 29° to 64°, thus supporting previous research indicating the change towards against-the-rule ocular astigmatism with age is due to corneal changes,55,58,59 which may be associated with an age-related reduction in eyelid tension.60,61
All objective measurements of refraction were acquired without the prior instillation of a cycloplegic agent. Non-cycloplegic refraction was chosen to ensure involvement in the study did not impair the ability of the cohort, who were mostly Aston University staff, to return to work immediately following the appointment. In addition, the discomfort associated with cycloplegic eye drop instillation and the transient effects may have discouraged participants from attending future review appointments, risking an unacceptably high attrition rate and reducing the power of the study. Nevertheless, the individuals who participated in the present investigation had relatively low amplitudes of accommodation, therefore, it is unlikely erroneous accommodation would have had a significant impact on the magnitude of the results in the current study.
At the conclusion of the 2.5-year study, participants who underwent a myopic shift in refraction >0.50D were invited to return for re-measurement of their objective refraction under cycloplegia. The aim of measuring cycloplegic refraction was to determine whether a spasm of accommodation, instigated by ciliary muscle contraction, might be responsible for the myopic shift in refraction. Unfortunately, it was not possible to obtain cycloplegic measurements from the 2 participants who experienced a myopic shift during the course of the present study due to narrow anterior chamber angles (van Herick62 grade 2) and failure to consent. As well as investigating differences between non-cycloplegic and cycloplegic refraction, future studies should also quantify longitudinal changes in ciliary muscle morphology and axial biometry to investigate the structural correlate responsible for the myopic shift in MSE during incipient presbyopia.
When reflecting on the limitations of the study, the addition of cycloplegic refractions and axial length data could provide further explanation for the refractive changes observed in the cohort. Additionally, the multi-ethnicity of the cohort will introduce some inhomogeneity; however, the vast majority of the participants (82%, see Table 1) were White European, so the influence of ethnic differences is likely to be limited.
The current study provides the first prospective, longitudinal insight into how refractive error progresses during incipient presbyopia. In conclusion, the incidence of a myopic shift in refractive error during incipient presbyopia does not appear to be as large as previously indicated by retrospective research.5,33 The incidence of a hypermetropic shift is greater than the myopic shift, however overall, a significant change in refraction is not evident over 2.5 years during incipient presbyopia. The structural origins of the hypermetropic and myopic shifts in refraction are unclear and require further investigation, particularly focused on the accommodative apparatus in order to determine whether a loss of accommodative ability is associated.
Conflicts of interestThe authors have no conflicts of interest to declare.
DL was funded by the College of Optometrists, UK. The work described herein was presented, in part, at the Association for Research in Vision and Ophthalmology (ARVO) annual conference (Laughton DS, et al. Invest Ophthalmol Vis Sci. 2014;55:ARVO E-Abstract 3744).