To investigate vestibulo ocular reflex (VOR) in MS patients without any history of optic neuritis.
Methods26 MS patients without any previous history of optic neuritis and 13 age- matched control subjects were included in this study. Their age ranged from 22 to 50 years old. We evaluated monocular visual evoked potential (VEP), monocular and binocular best corrected static and dynamic visual acuity, near and distance phoria and VOR gain.
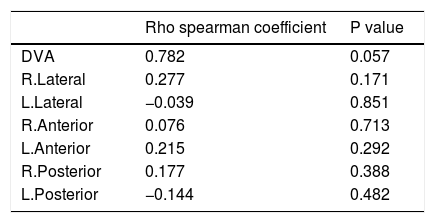
ResultsMean spherical equivalent (SE) was – 0.40 ± 0.93 D and – 0.04 ± 0.14 D for study and control group, respectively (P = 0.060). There was a significant difference in dynamic visual acuity (DVA) between two groups (P = 0.029). VOR gain was not significantly different in both groups through vHIT measurements (P = 0.338). Duration of MS had a mean of 78.38 ± 75.94 months (ranged from 6 to 336 months). We found no significant correlation between disease duration and VOR (Rho 0.277, P = 0.171) or DVA (Rho 0.782, P = 0.057).
ConclusionOur study showed that although vHIT results decreased in MS patients, there was no significant differences between two groups.
Multiple sclerosis (MS) is a progressive disease that affects central nervous system (CNS). Over 2.5 million people are suffering from MS in the world.1 As MS progresses, involvement of brainstem and cerebellar structures affect the eye motor system. Any defect in the eye motor system causes more advanced disability.2 Vestibulo ocular reflex (VOR) is an important component of the eye motor system that can stabilize the gaze during head movements.3 This mechanism causes compensatory eye movements to retain retinal image while head is moving fast.3 Among MS patients, 68–72% have brainstem lesions, 87% have abnormal brainstem reflexes and up to 70% have cerebellar symptoms.4 VOR function is processed in the central vestibular nuclear complex of the brainstem and then the cerebellum modulates it. VOR defects can be a preliminary sign of MS or become apparent later.5 The important point is that VOR dysfunction sometimes is reported as a visual symptom but if there is no optic nerve involvement along with normal brainstem magnetic resonance imaging (MRI), visual evoked potential (VEP) results will be normal.6 Any VOR abnormality affects the ability to keep the gaze stabilized on a target during head movements.7 In this situation, the gaze changes with head movements and there is a need of compensatory saccades for fixation on the target.7 Dynamic visual acuity (DVA) and video head impulse test (vHIT) are clinical tests to measure VOR.8 The aim of this study is to investigate VOR function in MS patients without a previous history of optic neuritis by measuring DVA and vHIT.
Material and methodsTwenty-six patients with definite diagnosis of MS referred by a neurologist were enrolled in this study. Inclusion criteria were definite diagnosis of MS according to 2010 McDonald corrected criteria,9 normal brainstem MRI, age of 20–50 years and best corrected visual acuity (BCVA) of 20/20 or better in both eyes. Exclusion criteria included the presence of any visual symptom, such as blur or loss of vision, diplopia and painful eye, ocular pathologic condition, nystagmus, strabismus and eye movement defect, systemic diseases, such as diabetes mellitus and hypertension, any history of vestibular system disease like Meniere’s disease and Labyrinthitis, neck muscle disorder and the use of drugs affecting the vestibular function.
At first, all demographic (sex and age) and MS-related variables (onset time, primary symptom, disease duration, history of optic neuritis and treatment) were recorded. The presence of previous optic neuritis was assessed using patients’ Electronical Medical Record, careful ophthalmoscope evaluation and VEP test. VEP was done according to the ISCEV standards,10 with two pattern reversal target size of 15 and 60 min of arc in frequency level of 1 Hz at 1-m distance. One hundred responses were summed from each eye tested separately. Then complete optometry examination was accomplished and DVA was measured binocularly in a manner of best-corrected static visual acuity (SVA) of 20/20 or better. To measure DVA clinically, the examiner swung the patient's head horizontally in a frequency of daily activities (approximately 2 Hz) to the right-left side at 20° angle, and asked the patient to read a visual acuity chart at the distance of 4 m. DVA was recorded in Log MAR.
The next step was VOR gain measurement using video head impulse test (vHIT). After putting and adjusting the goggles on the patient’s eyes, the examiner asked the patient to keep the fixation on a stable object and manipulated patient's head quickly and precisely.11 This test shows overt and covert saccades.
Declaration of interestThe study followed the tenets of the Declaration of Helsinki. All patients were appropriately informed before their participation in this study, and after a complete ophthalmic examination and a thorough discussion of the risks and benefits of the study, all participants gave written informed consent.
Statistical analysisStatistical analysis was performed using SPSS for Windows software (version 16, SPSS, IBM, New York, United states). Variables were expressed as the mean ± standard deviation. Shapiro–Wilk test was used to determine normality of each variable. Then, parametric tests were used for variables showing a Normal distribution and nonparametric tests for variables not following a Normal distribution. Associations between VOR gain and DVA outcomes with disease duration and first symptom type (whether sense or motor type) were also evaluated using the Spearman correlation analysis. Linear regression models were assessed to show different variable roles in predicting DVA and VOR gain impairment. Differences were considered statistically significant when the P value was 0.05 or less.
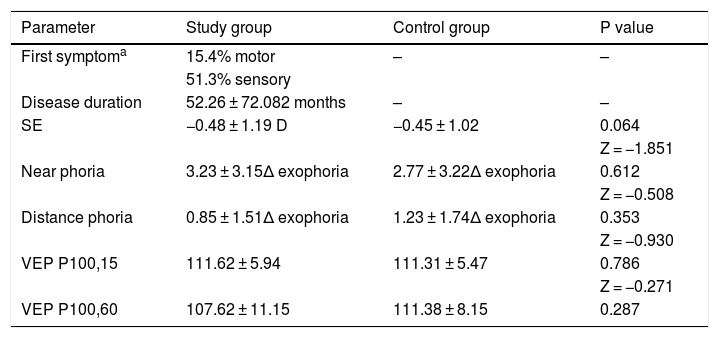
ResultsTwenty-six MS patients (6 males and 20 females), with mean age of 36.42 ± 9.39 years (range 22–50 years), and 13 healthy patients (2 males and 11 females), with mean age of 36.38 ± 10.37 years (range 22–50 years), participated in this study. Table 1 shows some characteristics of MS patients and control group eyes. Right eye values are presented.
MS patients’ characteristics.
| Parameter | Study group | Control group | P value |
|---|---|---|---|
| First symptoma | 15.4% motor | – | – |
| 51.3% sensory | |||
| Disease duration | 52.26 ± 72.082 months | – | – |
| SE | −0.48 ± 1.19 D | −0.45 ± 1.02 | 0.064 |
| Z = −1.851 | |||
| Near phoria | 3.23 ± 3.15Δ exophoria | 2.77 ± 3.22Δ exophoria | 0.612 |
| Z = −0.508 | |||
| Distance phoria | 0.85 ± 1.51Δ exophoria | 1.23 ± 1.74Δ exophoria | 0.353 |
| Z = −0.930 | |||
| VEP P100,15 | 111.62 ± 5.94 | 111.31 ± 5.47 | 0.786 |
| Z = −0.271 | |||
| VEP P100,60 | 107.62 ± 11.15 | 111.38 ± 8.15 | 0.287 |
All values are presented in mean ± standard deviation. SE = Spherical Equivalent, P100,15 = P100 for 15 min of arc target size, P100,60 = P100 for 60 min of arc target size.
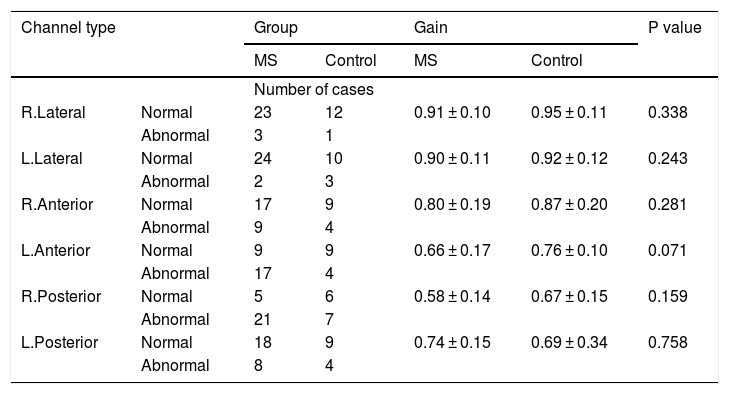
As shown in Table 1, there was no significant difference between the two groups. DVA measurements showed significant differences between patients and normal subjects (P = 0.029). VOR gains were also compared between two groups. Table 2 shows VOR gains of different channels measured by vHIT in both study and control groups.
Comparison of VOR gain between MS and control groups.
| Channel type | Group | Gain | P value | |||
|---|---|---|---|---|---|---|
| MS | Control | MS | Control | |||
| Number of cases | ||||||
| R.Lateral | Normal | 23 | 12 | 0.91 ± 0.10 | 0.95 ± 0.11 | 0.338 |
| Abnormal | 3 | 1 | ||||
| L.Lateral | Normal | 24 | 10 | 0.90 ± 0.11 | 0.92 ± 0.12 | 0.243 |
| Abnormal | 2 | 3 | ||||
| R.Anterior | Normal | 17 | 9 | 0.80 ± 0.19 | 0.87 ± 0.20 | 0.281 |
| Abnormal | 9 | 4 | ||||
| L.Anterior | Normal | 9 | 9 | 0.66 ± 0.17 | 0.76 ± 0.10 | 0.071 |
| Abnormal | 17 | 4 | ||||
| R.Posterior | Normal | 5 | 6 | 0.58 ± 0.14 | 0.67 ± 0.15 | 0.159 |
| Abnormal | 21 | 7 | ||||
| L.Posterior | Normal | 18 | 9 | 0.74 ± 0.15 | 0.69 ± 0.34 | 0.758 |
| Abnormal | 8 | 4 | ||||
R: right, L: left.
Although VOR shows better results in the control group, the difference between groups was not statistically significant. Associations between disease duration and patients’ first symptom presentation age with VOR and DVA were also assessed. Spearman correlation test results are presented in Tables 3 and 4.
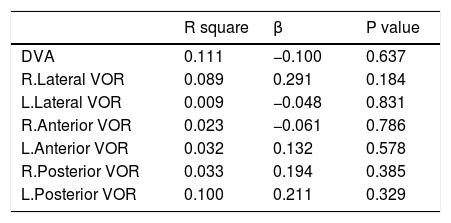
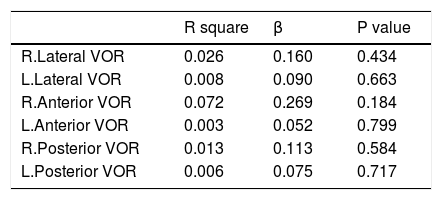
Linear regression analysis was used to assess the independent role of MS duration and DVA in the VOR gain impairment among MS patients with no history of optic neuritis. Age was considered as a confounder in these analysis. Regression test showed no significant linear relation between these parameters. Tables 5 and 6 show regression findings.
Linear regression analysis of VOR with MS duration.
| R square | β | P value | |
|---|---|---|---|
| DVA | 0.111 | −0.100 | 0.637 |
| R.Lateral VOR | 0.089 | 0.291 | 0.184 |
| L.Lateral VOR | 0.009 | −0.048 | 0.831 |
| R.Anterior VOR | 0.023 | −0.061 | 0.786 |
| L.Anterior VOR | 0.032 | 0.132 | 0.578 |
| R.Posterior VOR | 0.033 | 0.194 | 0.385 |
| L.Posterior VOR | 0.100 | 0.211 | 0.329 |
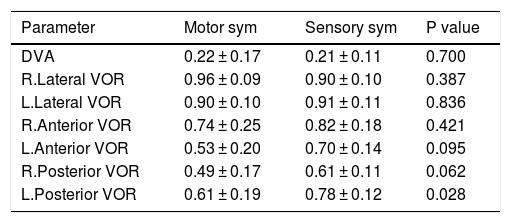
Comparison between the type of primary symptoms (whether sensory or motor) of the patient at the onset of disease and their results of DVA and VOR was also assessed [Table 7].
Comparison between different primary symptom at the onset of MS.
| Parameter | Motor sym | Sensory sym | P value |
|---|---|---|---|
| DVA | 0.22 ± 0.17 | 0.21 ± 0.11 | 0.700 |
| R.Lateral VOR | 0.96 ± 0.09 | 0.90 ± 0.10 | 0.387 |
| L.Lateral VOR | 0.90 ± 0.10 | 0.91 ± 0.11 | 0.836 |
| R.Anterior VOR | 0.74 ± 0.25 | 0.82 ± 0.18 | 0.421 |
| L.Anterior VOR | 0.53 ± 0.20 | 0.70 ± 0.14 | 0.095 |
| R.Posterior VOR | 0.49 ± 0.17 | 0.61 ± 0.11 | 0.062 |
| L.Posterior VOR | 0.61 ± 0.19 | 0.78 ± 0.12 | 0.028 |
As Table 7 shows, VOR gain was lower for patients who had motor symptoms at the onset of MS, but the difference was not statistically significant.
DiscussionIn this study, no statistically significant correlations were found between VOR outcomes and MS duration and patients’ first symptom at the onset of disease in MS patients with no prior history of optic neuritis and normal brainstem in MRI results. When controlling for age, none of these variables were independently associated with higher probability of VOR dysfunction. Our results suggest that VOR function is independent of disease duration.
The visual pathway is highly susceptible to damage in MS. This explains the relevance of visual impairment in MS, being the second cause of life quality loss in this type of patients. VOR is an important part of the visual function. Both VOR dysfunction and optic neuritis have visual symptoms,1 so if a MS patient has these symptoms without any definite diagnosis of optic neuritis, VOR can also be tested. In this study, VOR was assessed using vHIT gain and DVA to assess both physiological and non-physiological VOR, respectively. DVA test is considered behavioral (and not physiological) because correct identification and report of optotype orientation drive test scoring instead of direct measures of eye movement response kinematics. As DVA can be affected by other common dysfunctions in MS patients, such as coordination, motor learning and others, another method to measure physiological VOR was performed.12
Both DVA and vHIT tests showed reductions in MS patients compared with the control group. These results were observed under normal brainstem in MRI results. DVA was significantly lower in MS patients, but VOR reduction was not statistically significant. This can be justified by the presence of other common dysfunctions in MS patients that can interfere in DVA results as a behavioral test. This is a point that was previously noted by Odom et al.10 This is in agreement with Habek2 who demonstrated that brainstem dysfunctions may be even available in MS patients with normal brainstem showed in MRI. As Pavlović et al.13 have noted, vHIT can be used as a diagnostic test for brainstem lesions so any decrease in vHIT results may predict the start of brainstem involvement in MS patients. Therefore, it seems to be necessary to include a VOR function test during clinical practice of MS patients.
As there was no history of optic neuritis and brainstem lesion, no significant auditory and eye motor dysfunction was expected. Other similar studies1,14,15 had no filtering based on parameters, such as brainstem lesion and optic neuritis that can influence VOR function.
Patient’s primary symptom at the beginning of the MS in the form of motor or sensory symptom16 was also considered. Patients were divided into two groups according to their primary symptom at the MS onset. Any symptom such as muscle weakness or heaviness, loss of hand proficiency, unsteady walking and spasticity was set in the motor group17 and fatigue, depression, blur vision, halos in the visual field and double vision was considered as sensory group.17 Analysis between MS patients showed that patients with primary motor symptom had lower results in anterior and posterior VOR gain. This can be explained in a way that MS patients with primary motor symptoms at the onset of their disease may be more susceptible to have earlier brainstem lesions and VOR dysfunction.
Our study had some limitations. First, it was better to use other electrophysiological tests in the evaluation of brainstem such as Evoked Potentials (EP) and Vestibular Evoked Myogenic Potentials (VEMP) to compare VOR deficiencies. Second, our samples were small and it is presented as a pilot study. Similar future studies with more sample participation can result in more definite improvement in clinical protocols.
Ultimately, it will be useful to compare VOR function among MS patients with and without prior history of optic neuritis.
ConclusionIn conclusion, even if MRI shows normal brainstem results and there is no history of optic neuritis, VOR can be checked as a supplementary test in MS patient. Although vHIT and DVA are two common methods for investigating VOR, vHIT is better and more reliable as a measure of physiologic VOR.
Conflicts of InterestThe authors have no conflicts of interest to declare.
FundingThis research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.